Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Revista de la Facultad de Ciencias Veterinarias
versión impresa ISSN 0258-6576
Rev. Fac. Cienc. Vet. vol.55 no.1 Maracay jun. 2014
The Stomach of the Adult African Catfish (Clarias gariepinus, Siluriforme s: Clariidae) in Farm Conditions: A Morphological and Mucin Histochem istry Analysis
El Estómago del Bagre Africano Adulto (Clarias gariepinus, Siluriformes: Clariidae) en Condiciones de Granja: Análisis Morforlógico e Histoquímico por Mucina
Ikpegbu Ekele*,1, Nlebedum Uchenna*, Nnadozie Okechukwu* and Agbakwuru Isaiah*
*Department of Veterinary Anatomy, Michael Okpara University of Agriculture, Umudike, Abia State, Nigeria
1 A quien debe dirigirse la correspondencia (To whom correspondence should be addressed)
Correo-E: fikpegbu@yahoo.com
Abstract
The stomach of the farmed African catfish (Clarias gariepinus, Siluriformes: clariidae) was investigated in this study to establish its basic anatomy, as there is scanty information available on the literature. It produced to baseline data for further investigative research, help fish clinicians in pathology and aid nutritionists in feed management. After cropping from a commercial aquaculture, apparently healthy fish were immobilized and euthanized by using chloroform. The samples were dissected and slices of cardiac, fundic and pyloric stomach processed through routine histological procedures. Grossly, the stomach was J-shape. Cranial to the stomach was the oesophagus but no sphincter was seen separating them. Caudally, a pyloric sphincter separated the stomach from the proximal intestine. The stomach contained three regions cardiac, fundic and pyloric. The entire stomach surface was lined by a simple columnar epithelium containing mucin at the apical cytoplasm. Only the cardiac and fundic regions contained gastric glands in the lamina propria, but the glands were more developed in the fundic region. The tunica muscularis contained smooth muscle cells in an inner circular and outer longitudinal orientation. Mucin histochemistry revealed the presence of only neutral mucin in the stomach. The results obtained in this study suggest that the absence of gastric glands in the pylorus may be an adaptation of this species to reduce the quantity of acid entering the proximal intestine; hence it might help the alkaline medium to maximize pancreatic enzyme actions in the proximal intestine. The presence of only neutral mucin in the stomach will act as a buffer to neutralize the high acidic stomach content. Neutral mucins are also associated with transport of macromolecules.
(Key words: Culture; animal morphology; mucin; histochemistry; enzymes; animal glands; stomach; african catfish)
Resumen
Se efectuó un estudio de tipo histológico e histoquímico con mucina, en el estómago del bagre africano (Clarias gariepinus, Siluriformes: clariidae) cultivado en granja acuícola, para establecer su anatomía básica, debido a la escasa información disponible en la literatura. La investigación generó datos para ulteriores investigaciones, que contribuirán con los ictiopatólogos y ayudarán a los nutricionistas en el manejo alimentario. Después de su cultivo en una granja acuícola comercial, los peces aparentemente sanos, fueron inmovilizados y sacrificados, usando cloroformo. Se tomaron muestras de las regiones cardial, fúndica y pilórica, y se procesaron histológicamente. En su conjunto, el estómago presentaba una apariencia de J. El esófago estaba situado cranealmente al estómago, pero no se observó un esfínter que los separara. Caudalmente, un esfínter pilórico separaba al estómago del intestino proximal. El estómago contenía tres regiones: cardiaca, fúndica y pilórica. La totalidad de la superficie estomacal se encontraba recubierta por un epitelio cilíndrico, que contenía mucina en el citoplasma apical. Solamente las regiones cardiaca y fúndica tenían glándulas gástricas en la lámina propia, que eran más desarrolladas en la región fúndica. La túnica muscular poseía células musculares lisas con una orientación interna circular y externa longitudinal. La histoquímica mediante la mucina reveló la presencia solo de mucina neutra en el estómago. Los resultados obtenidos en este estudio sugieren que la ausencia de glándulas gástricas en el píloro, pudiera ser el resultado de una adaptación de esta especie para reducir la cantidad de ácido que penetra en el intestino proximal; por tanto, pudiera ayudar al medio alcalino a maximizar las acciones de las enzimas pancreáticas en el intestino proximal. La sola presencia de mucina neutra en el estómago actuaría como amortiguador contra el elevado contenido ácido del estómago. Las mucinas neutras también están asociadas con el transporte de micromoléculas.
(Palabras clave: Cultivo; anatomía animal; mucina; histoquímica; enzimas; glándulas animales; estómago; bagre africano)
Recibido: 23/09/13 - Aprobado: 03/07/14
Introduction
The stomach is a dilated organ where the oesophagus opens into. When present, it constitutes a criterion to classify teleost into gastric and agastric groups. The stomach shapes and sizes of various studied bony fishes have also been documented and are associated with the duration between meals and nature of food [1]. The three stomach regions: cardiac, fundic and pylorus, with various degrees of gastric gland development have been reported in the literature [2-6]. It has also been stated that the teleost stomach wall comprises the layers typical of vertebrate digestive tract in addition to extra features like stratum compactum and stratum granulosum. Distinct muscularis mucosae have been documented [7, 8]. One type of secretory cell, the oxyntopeptic cell, producing both pepsinogen and hydrochloric acid, has been reported in most teleost gastric gland [5, 9, 10].
The gastric mucosa is believed to perform an absorptive function. This function is suggested by the presence of numerous blood vessels in the lamina propria of Peristedium longispatha pyloric stomach [11] and the cardiac stomach of Mugil tade F. [12].
Despite extensive investigation of most teleost in reported literature, there is dearth of information in available literature on the morphology of the African catfish stomach from Nigerian commercial farms, hence the need for this study. The knowledge derived from this study will help fish nutritionists and pathologists.
Materials and Methods
Ten adult African catfish sourced from a commercial aquaculture in Eastern Nigeria were used for the study. They average weighed was 900 g and the body length was 45cm. The fish were euthanized with chloroform. The body cavity was cut open through the ventral surface and the alimentary tract dissected out. The stomach of the specimen under study (Figure 1) was excised and sections of cardiac, fundic and pyloric regions were immediately fixed in 10% neutral buffered formalin for 48 h.
The tissues were passed through graded ethanol, cleared in xylene, impregnated and embedded in paraffin wax. Sections of 5-6 µm thick were obtained using a Leitz microtome model 1512. Tissues were stained with haematoxylin and eosin for light microscopy examination [13]. The presence of mucins was demonstrated using alcian blue (AB) at pH 2.5 [14,15] and the periodic acid of Schiff (PAS) procedure with and without prior digestion with diastase [16,17]. In addition, the PAS technique was employed in combination with AB for neutral and acid mucin [13]. Photomicrographs were taken with a Motican 2001 camera (Motican, UK) attached to an Olympus microscope.
Results
Grossly, the stomach was J-shaped. The small arm of the J, the most caudal part was occupied by the cardiac region, and the fundic region formed about four-fifth of the long arm of the J or the middle part, while the pylorus occupied the tip of the J-shaped stomach and was the most cranial part of the stomach (Figure 1).The pyloric end was somewhat ventral to the gastro-oesophageal opening. The pyloric region was cranial, while the fundus was the caudal region of the stomach. The pylorus was separated from the proximal intestine by a constriction (Figure 2).
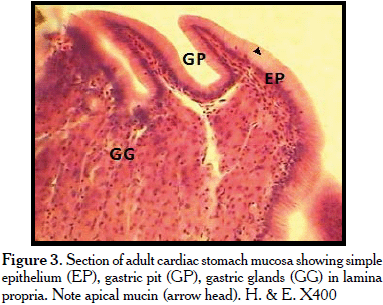
Histologically, the cardiac stomach epithelium covering was of simple columnar epithelium. Each individual columnar cell had a basal nucleus and mucin in the apical cytoplasm. The epithelium also contained intraepithelial leukocytes (Figure 3). Gastric pits were also seen (Figure 3). The lamina propria contained gastric glands; beneath the glands were collagen connective tissue fibres. The tunica muscularis contained smooth muscle cells in an inner circular and outer longitudinal orientation. Myenteric plexus was observed between the two muscle layers. Tunica serosa was observed. Subserosal blood vessels were also seen. The fundic stomach was covered by simple columnar mucous epithelium with apical mucin. The gastric pits were narrower and deeper than those observed in the cardiac stomach (Figures 4, 5). The lamina propria contained larger gastric glands than the cardiac stomach (Figures 4, 6). The pyloric stomach was also covered by a simple mucous epithelium. The gastric pits were very shallow. The lamina propria contained no gastric glands (Figure 7). Histochemically, the apical mucin in the simple columnar epithelium was positive to PAS (Figures 9,10), negative to AB (Figure 11), when subjected to combined AB and PAS procedures, and only neutral mucin was revealed (Figures 12,13).





Discussion
Grossly, the stomach was J-shaped. This shape has been reported in Oncorhynchus mykiss [2] and may help extend the time feed stays in the organ, thereby ensuring greater degree of digestion by gastric enzymes. An U-shaped stomach has been reported in Salmo, Coregonus and Clupea [1], Glyptosternums maculatum [18]; The Y-shape stomach has been reported in Oreochromis niloticus [3], Micropogonias furnieri [5], and the straight stomach in Esox [1]. The cardiac, fundic and pyloric regions were seen as reported in some teleosts [18]. The general microanatomy of the regions was similar, except for the absence of gastric glands in the pylorus, which may be attributed to the need for reduced gastric acid secretion that may interfere with the needed alkaline medium for digestive processes in the intestine by pancreatic enzymes [19]. Also, this pyloric stomach like in most teleosts stomach was characterized by absence of gastric glands, well developed thick tunica muscularis comprising mainly circular muscle. The reduced submucosa with collagen fibres is a modification to serve the gizzard to triturate and mix [5, 20, 21].
The mucosal folds seen in the regions were highest in fundic stomach but shallowest in the pyloric region. The cardiac stomach was in-between. This has been reported in the walking catfish Clarias batrachus [6], and may be related to the development of the gastric glands.
The gastric epithelium was of simple columnar absorptive type with apical mucin. No goblet cells were observed. This finding is supported by previous studies seen in literature [2,10, 22-25]. In the bay snook Petenia splendida (Gunther), the gastric mucosa was lined by simple cuboidal epithelium [26]. The apical region of the columnar epithelia containing mucin is involved in preventing auto- digestion and reducing the effect of gastric acid content [2, 24, 26, 27].
The location of gastric glands is species – specific and has been related to feeding habits. In this study, the gastric glands were seen only in cardia and fundus. Absence of gastric gland in pylorus has been reported by Zacarias et al. [28] in their study of the Paralichtlys californicus larvae. Also, Yang et al. [10] in Pelteobagrus fulvidraco and Xiong et al. [18] in Glyposternum maculatum reported the same findings. In Diplodus puntazzo, Pleuronectes ferruginea, summer flounder – Paralichthys dentatus, the fundic region is the only glandular region. In Diplodus sargus, Umbrina cirrosa and Paudoro pagrus only the cardiac region was seen to be glandular. On the other hand, in Pandora pagellus the entire stomach is glandular [25]. The Oreochromis niloticus stomach is entirety glandular [3]. Only the presence of gastric glands in the cardias and fundus has also been reported by Chen et al. [29] in their work on Kingfish Seriola lalandi. They also reported the presence of PAS positive apical borders of the epithelia as seen in this study. The fundic region was described by them, to be the largest region of the stomach. This report supports our findings where the fundus formed the base and two-thirds (2/3) of the J-shaped stomach; and this may be related to the level of development of gastric glands in this region and the need for more contact time between feed and the secreted enzymes since this species is an omnivore. Cao and Wang [21] reported the absence of gastric glands in the pylorus in the study of yellow catfish.
The cells of the gastric glands will probably be oxyntopeptic cells [9, 10, 18 30] producing both pepsinogen and hydrochloric acid, but some researchers have reported two separate cell types: mucous cell and another one with zymogen granules [21], but further investigation should be done to ascertain this claim. The fibrocytes may be producing collagen to surround gastric glands in their lobules or the cells may be myoepithelial cell producing contractile fibres that will help elaborate the secretory products of the oxyntopeptic cells, but further investigation is required to verify the nature and function of these cells.
The collagen bundle in the submucosa and thick tunica muscularis smooth muscles may be for supporting and strengthening since stratum compactun is lacking in this species [31]. The dense smooth muscle layer seen in the pyloric sphincter has been reported and involuntarily controls rate of food passage, affecting retention time and volumetric capacity of the stomach [4, 32]. These dense circular smooth muscles in the sphincter may also be involved in trituration [20, 21]. The presence of skeletal muscle in pyloric sphincter has been reported [3] and is associated with voluntary control over food entry into the intestine.
The inner circular and outer longitudinal smooth muscle layers of the tunica muscularis of the stomach observed have been reported and is involved in involuntary contraction for food digestion [4,5,18]. The myenteric plexus seen is for nerve supply and stimulation [2, 4]. Presence of serosa consisting of mesothelial cells and loose connective tissue containing small blood vessels as seen in this study has been reported [33].
Mucin histochemistry revealed that only neutral mucin was seen in the stomach. This is consistent with previous findings in most teleosts [5, 6, 21, 24, 26]. The presence of only neutral mucin on the stomach epithelia has been suggested to have a role in transport of macromolecules, increase in digestive efficiency, to defend against bacteria, buffering effect on high acidity of stomach content this exerting a preventing proteolytic damage to epithelium [25-27, 34, 35].
The presence of acid mucin in the stomach has been reported in Umbrina cirrosa and is associated with prevention of damage to gut epithelium, acting as lubricant to fibre-rich materials [27]; also, acid mucin has been reported in Oreochromis niloticus [3, 32].
Reference
1. Smith LS. Digestion in Teleost fishes. FAO Corp Doc Repository. 1978; 1-19. [ Links ]
2. Ezeasor DN. The fine structure of the gastric epithelium of the rainbow trout, Salmo gairdneri, Richardson. J Fish Biol. 1981; 19:611-27. [ Links ]
3. Caceci T, El-Habback HA, Smith SA, Smith BJ. The stomach of Oreochromis niloticus has three regions. J Fish Biol. 1997; 50:939-52. [ Links ]
4. Albrecht MP, Ferreira MFN, Caramasch EP. Anatomical features and histology of the digestive tract of two related neotropical omnivorous fishes (Characiformes; Anostomidae). J Fish Biol. 2001; 58: 419- 30. [ Links ]
5. Diaz AO, Garcia AM, Figueroa DE, Goldemberg AL. The mucosa of the digestive tract in Micropogonias furmieri: A light and Electron microscope approach. Anat Histol Embryol. 2008; 37:251-56. [ Links ]
6. Raji AR, Norouzi E. Histological and histochemical study on the alimentary canal in walking catfish (Clarias batrachus) and piranha (Serrasalmus nattereri). Iran J Vet Res Shiraz Univ. 2010; 11:255-61. [ Links ]
7. Khanna SS. Histology of the digestive tract of a teleost, Clarias baltrachus (L). J Zoo Soc of India. 1964; 16:53-8. [ Links ]
8. Bishop C, Odense PH. Morphology of the digestive tract of the cod, Gadus morhua. J Fish Resh Board of Canada. 1966; 23:1607-615. [ Links ]
9. Barrington EJW. The alimentary canal and digestion. In the physiology of fishes (M.E. Brown, ed), Vol. 1, Ch.3. 1957; p. 109-61. [ Links ]
10. Yang R, Xie C, Fan Q, Gao C, Fang L. Ontogeny of the digestive tract in yellow catfish Pelteobagrus fulvidraco larvae. Aquaculture. 2010; 302:112-23. [ Links ]
11. Chan VM. The histology of the alimentary tract of the deep-water gurnard Perisledious longispatha (Goode and Bean). Univ Neb Std Linc. 1941; 41:7-30. [ Links ]
12. Pillay TVR. Studies on the food, feeding habits and alimentary tract of the grey mullet, Mugil tade forskal. Proced of the Nat Inst Sci India. 1953; 19:777-827. [ Links ]
13. Bancroft JD, Stevens A. Theory and practice of histological techniques. (Churchill Livingstone, New York, USA. 1977; p. 148-155. [ Links ]
14. Steedman HF. Alcian blue 8G: a new stain for mucin. J Micro Sci. 1950; 91:477-79. [ Links ]
15. Lev R, Spicer SS. Specific staining of sulphated groups with alcian blue at low pH. J Histochem Cytochem. 1964; 12:309-310. [ Links ]
16. Lillie RD, Greco J. Mact diastase ptyalin in place of saliva in the identification of glycogen. Stain Tech. 1947; 22:67-70. [ Links ]
17. Ikpegbu E, Nlebedum UC, Nnadozie O, Agbakwuru I. Fast Green FCF or Ehrlichs hematoxylin as counterstain to periodic acid Schiff reaction: A comparative study. Histologic 2011; 54:29-30. [ Links ]
18. Xiong D, Zhang L, Yu H, Xie C, Kong Y, Zeng Y, Huo B, Liu Z. A study of morphological and histology of the alimentary tract of Glyptosternum maculatum (Sisoridae, silurifores). Acta Zool. 2011; 96:161-69. [ Links ]
19. Ribeiro L, Sarazquete C, Dinis MT. Histological and histochemical development of the digestive system of Solea senegalensis (Kaup, 1858) larve. Aquacul. 1999; 171:293-308. [ Links ]
20. Schmitz EH, Baker CD. Digestive anatomy of the gizzard shad, dorosoma cepedianum and the threadfin shad, D. petenens. Trans Ameri Mic Soc. 1969; 88:525-46. [ Links ]
21. Cao JX, Wang WM. Histology and mucin hstochemistry of the digestive tract of Yellow Catfish Petteobagrus fulvidraco. Anat Histol Embryol. 2009; 38:254-61. [ Links ]
22. Sarieyyupoglu M, Girgin A, Koprucu S. Histological study in the digestive tract on larval development of rainbow trout (Oncorhynchus mykiss, Walbaum, 1792). Truk J Zool. 2000; 24:199-205. [ Links ]
23. Hamlin HJ, Hunt von Herbing I, Kling LJ. Histological and morphological evulation of the digestive tract and associated organs of haddock throughout post – hatching ontogeny. J Fish Biol. 2000; 57:716-32. [ Links ]
24. Kozaric Z, Kuzir S, Petrinec Z, Gjurevic E, Bozic M. The development of the digestive tract in larval European Catfish (Silures glanis L.). Anat Histol Embroyol. 2008; 37:141-46. [ Links ]
25. Micale V, Mughia U. Comparative ontogeny of the digestive tract in Sharpsnout sea Bream Diplodus puntazzo C. and common Pandora Pagellus enthrinus L. Open Mar Biol J. 2011; 5:31-4. [ Links ]
26. Trevino L, Alvarez-Gonzalez CA, Perales-Garcia N, Avevalo-Galan L, Uscanga-Martinez A, Morquez-Couturier G, Fernandez I, Gisbert E. A histological study of the organogensis of the digestive system in baysnook (Peternia splendid Gunther, 1862) from hatching to the juvenile stage. J Appl Ichthyol. 2011; 27:73-82. [ Links ]
27. Pedini V, Scocco P, Radaelhi G, Fagioli O, Ceccarelli P. Carbohydrate histochemistry of the alimentary canal of the Shi. Drun, Umbrina Cirrosa. Anatomia Histologia Embryologia. 2001; 30:345-49. [ Links ]
28. Zacarias-Soto M, Muguet JB, Lzao JP. Proteolytic activity in California Habitat larvae (Paralichthys californiaus) J World Aquacul Soc. 2006; 37:175-85. [ Links ]
29. Chen BN, Qin JG, Kumar MS, Wayne H, Clarke S. Ontogenetic development of the digestive system in yellowtail Kingfish Seriola lalandi Larvae. Aquacul. 2006; 256:489- 501. [ Links ]
30. Verma SR, Tyagi MP. A comparative histological study of the stomach of a few teleost fishes. Gegen. Morph Jah. 1974; 120:244-53. [ Links ]
31. Burnstock G. The morphology of the gut of the brown trout (Salmo trutta). Quart J Micro Sci. 1959; 100:183-98. [ Links ]
32. Morrison CM, Wright Jr. A study of the histology of the digestive tract of the Nile tilapia. J Fish Biol. 1999; 54:597-606. [ Links ]
33. Arellano JM, Storch V, Sarasquete C. Histological and histochemical observations in the stomach of the Senegal sole Solea senegalensis Histol Histopathol. 2001; 16:511-21. [ Links ]
34. Scocco P, Menghi G, Ceccarelli P. Glycohistochemistry of the Tilapian spp. stomach. J Fish Biol. 1996; 49:584-93. [ Links ]
35. Elbal MT, Garcia Hernandez MP, Lozano MT, Agulleiro B. Development of the digestive tract of gilthead sea bream (Sparus aurata L.). Light and electron microscopic studies. Aquacul. 2004; 234:215-38. [ Links ]