Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Archivos Venezolanos de Farmacología y Terapéutica
versión impresa ISSN 0798-0264
AVFT vol.34 no.1 Caracas mar. 2015
Effectiveness and tolerability of the Petasites hybridus leaf extract Ze 339 in the treatment of allergic rhinitis in a paediatric population
Efectividad y tolerabilidad del extracto de la hoja de Petasites hybridus Ze 339 en el tratamiento de la rinitis alérgica en una población pediátrica
Cordula Molla*, Catherine Zahneraa, Mercedes Rodríguez de Marquisb, Maria González Yibirínb and Jairo J. Ruiz Rodríguezb, on behalf of the Study Group1
a Max Zeller Söhne AG, Romanshorn, Switzerland
b Laboratorios Leti, S.A.V., Guarenas, Venezuela
*Correspondence to Dr. C. Moll, Max Zeller Söhne AG, Romanshorn, Switzerland; E-mail: cordula.moll@zellerag.ch
1 Participating practical physicians are all listed in Rodríguez de Marquis & González Yibirín (2012).
Abstract
Background: Allergic rhinitis (AR) is one of the most prevalent chronic allergic diseases in children, with a high impact on a childs quality of life and co-morbidities like asthma.
Objective: The objective of this analysis was to investigate the effectiveness and tolerability of the Petasites hybridus leaf extract Ze 339 (Ze 339) in the treatment of AR in paediatric patients.
Methods: We present a paediatric sub-analysis of a recent Venezuelan observational study, which investigated the effect of Ze 339 on clinical symptoms of AR in patients treated under conditions of daily practice. Among those 927 previously studied patients, 92 patients were less than 18 years old. Thereof, we included 53 children and adolescents in this intention-to-treat sub-analysis. Patients were advised to take one tablet of Ze 339 (corresponding to 8 mg petasins) two or three times a day up to one month. Symptoms of AR were recorded at every medical visit using a 4-point rating scale to indicate the level of severity. Single symptoms of AR (rhinorrhoea, nasal congestion, sneezing, itchy eyes, red eyes, itchy throat) and several composite scores (total symptom score, total nasal symptom score, total nasal and ocular symptom score) were evaluated. The overall therapeutic response of patients to the medication indicating effectiveness and tolerability was evaluated by a 5-point rating scale by both the physician and patient.
Results: Full recovery and a significant improvement of symptoms were seen in 86.8% of patients. This was also reflected in a significant improvement of the different composite scores (p<0.001). Overall, the results showed that Ze 339 is effective and well tolerated in the treatment of AR in paediatric patients.
Conclusion: Ze 339 is effective and well tolerated in the relief of all symptoms of AR. Beneficial effects were reported by patients and physicians. Ze 339 may be considered for the treatment of AR in paediatric patients.
Keywords: Petasites hybridus, allergic rhinitis, children, adolescents, paediatric population, Ze 339
Recibido: 20/10/2015 Aceptado: 21/11/2015
Introduction
Traditionally, allergic rhinitis (AR) has been classified by the time and mode of exposure into three groups: seasonal, perennial, and more recently, the occupational one. The seasonal AR is caused by pollen from grass, weeds and trees. The perennial AR is caused in most cases by dermatophagoides mainly by excreta of house dust mites, animal dander, some fungi and cockroach. The occupational AR is caused by airborne agents in relation to work. It may be caused for example by laboratory animals, wood dust, grain or chemicals. According to the Allergic Rhinitis and its Impact on Asthma (ARIA) guideline1 AR is alternatively classified into two further broad categories regarding their time-course in intermittent and persistent AR. The duration of intermittent AR is less than 4 days a week and less than 4 consecutive weeks and persistent AR means that the symptoms are present more than 4 days a week and for more than 4 consecutive weeks. This classification of the ARIA guideline is more practical and appropriate to daily practice as the duration of AR has a high impact on the patients quality of life. Furthermore, AR is classified by severity and categorised into mild, moderate and severe based on individual symptoms (e.g. sleep disturbance, impairment of daily activities, leisure and/or sport, impairment of school or work)1.
As not only daily activities but also performance in school can be influenced by AR an effective treatment of AR is very important for children and adolescents. Both, the family physician and different specialists (allergologists, otolaryngologists, pulmonologists, paediatricians, etc.), are often involved in the treatment of patients with AR. The diagnosis is based primarily on clinical history, physical examination and allergic skin tests. Currently there are different therapeutic options for AR available. Dependent on the kind of symptoms, antihistamines, corticosteroids, leukotriene antagonists or cromoglycate are used. Since symptoms like rhinorrhoea and sneezing are histamine driven symptoms, antihistamines might be the treatment of choice for these symptoms. However, if the patient also suffers from nasal congestion, corticosteroids should be considered as additional treatment. Synthetic drugs for the treatment of AR show a good efficacy for relieving the symptoms. But for antihistamines (mainly those of the first generation) side effects like sedation are well known. Beside synthetic drugs there is also an effective and well tolerated herbal medicinal product containing the Petasites hybridus leaf extract Ze 339 (Ze 339) available for the treatment of AR. Ze 339 is a CO2-extract from leaves of Petasites hybridus, L. and standardized to 8 mg petasins (Drug-extraction ratio of 50- 100:1). The herbal medicinal products containing Ze 339 are indicated for the treatment of AR in patients from 12 years on. In clinical studies it was shown that Ze 339 relieves symptoms of AR like rhinorrhoea, sneezing, itchy nose and eyes and nasal congestion2-5. Ze 339 acts by various ways. Petasin and its isomers are considered to be the main active compounds of Ze 339 defining the anti-inflammatory activity of Ze 3394,6,7. Mast cells and eosinophils are two very important cells in the allergic process as their degranulation releases different mediators of AR like histamine and leukotrienes that cause the symptoms and typical complications of allergy. Ze 339 has an impact on the early response of AR by inhibiting mast cell and eosinophil degranulation, and on the late response of AR, the inflammation, by blocking leukotrienes and prostaglandins synthesis4,6,8,9. Therefore, a dual mode of action is suggested for Ze 339 due to its antiallergic and anti-inflammatory properties in the early and late phase of AR.
As paediatric patients are a special population, data from post marketing surveillance studies are of high interest to confirm the effectiveness and tolerability of Ze 339 in this special patient population. Three post marketing observational studies were published that investigated the treatment of AR with Ze 339. In these studies the inclusion of patients was not restricted to adults10-12. However, so far no detailed analysis of the paediatric population was published.
Objective
The objective of this sub-analysis from an observational study was to investigate the effectiveness and tolerability of Ze 339 in the treatment of AR in paediatric patients (patients aged from 2 to 18 years).
Methods
We evaluated the evolution of clinical symptoms of AR in patients treated with Ze 339 (Tesalin®; standardized to 8 mg of total petasins per tablet; manufactured by Zeller Medical AG Romanshorn, Switzerland) during an open prospective, postmarketing observational study of daily practice conditions in Venezuela12. The focus of the study was to evaluate the effectiveness and tolerability of Ze 339 in the Venezuelan population. According to the current law in Venezuela this kind of observational studies has not to be notified to the health agency and no approval by an ethics committee was necessary. The parents or the legal representatives signed a written informed consent for the use of their childrens study related data before participation. The sample included patients of any gender or ethnicity. Patients were informed about the therapy, advised to comply with the treatment and to attend medical checks. The results of the observational study including all patients (a total of 927 subjects) irrespectively of their age were previously published by Rodríguez de Marquis and González Yibirín12, however, no subgroup analysis of paediatric patients was performed.
In this paper, we therefore investigated the subset of paediatric patients. Among the total of 927 patients in the observational study, 92 patients were less than 18 years old and were included in the current sub-analysis. Patients, who attended at least one post-treatment follow-up consultation, were included in the intention-to-treat (ITT) analysis resulting in a study population of 53 children and adolescents (Figure 1).
In the original study, including 927 subjects, patients were excluded with a significant history of metabolic, cardiovascular, neurological, haematological, hepatic, gastrointestinal, cerebrovascular, respiratory or renal disease, or any other disorder, which the physician deemed likely to interfere with the study, or requiring any treatment likely to interfere with the objectives of the study. Furthermore, patients were excluded if they suffered from upper respiratory tract infections or sinusitis and would have required antibiotic therapy in the preceding two weeks, or if they experienced a viral respiratory infection during the week prior to the start of the study. Patients with suspected or clinical evidence of nasal candidiasis were also excluded12.
The symptoms of AR were recorded at each of their medical visits at the beginning of the treatment and on a follow up visit up to one month after starting treatment using a 4-point rating scale (0=absent, 1=mild, 2=moderate, 3=severe) to indicate the grade of severity12. In this sub-analysis further composite scores have been added. Beneath the evaluation of single symptoms (rhinorrhoea, nasal congestion, sneezing, itchy eyes, red eyes, itchy throat) also a total symptom score (TSS; sum score of all single symptoms), a total nasal symptom score (TNSS) with the three nasal symptoms (rhinorrhoea, nasal congestion, sneezing) and a total nasal and ocular symptom score (TNOSS) with the nasal symptoms and the symptoms itchy eyes and red eyes were evaluated according to the U.S. Food and Drug Administration Guidance for Industry on clinical trials for AR13. The overall therapeutic response of patients to the medication was evaluated by a 5-point rating scale for the improvement of symptoms (1=full recovery, 2=significant improvement, 3=moderate improvement, 4=mild improvement, 5=treatment failure). Based on this 5-point rating scale the effectiveness and tolerability of Ze 339 were analysed by both the physician and parents.
Patients took one tablet of Ze 339 two or three times a day for a maximum of one month. Furthermore, the patients were allowed to take any other concomitant medication for AR (including antihistamines, corticosteroids) and other, except antibiotic agents.
The IBM SPSS software (version 21.0) was used for statistical analyses. For analysis of demographics and improvement of symptoms descriptive statistics were used. For variables not normally distributed appropriate nonparametric tests were applied. The Wilcoxon signed ranks test was used for within-group comparison, the Mann-Whitney-U test for unpaired between-group comparisons and the Fishers exact test for contingency tables with small sample sizes. The level of significance was p=0.05.
Results
Patient characteristics
The overall results of this observational study were published by Rodríguez de Marquis and González Yibirín12 and out of 927 subjects 92 patients were less than 18-years-old defining the paediatric population. Thereof, 53 patients were included into the ITT population of this sub-analysis (Figure 1). In the latter population, four patients were children (median=10.5 years) and 49 patients were adolescents (median=15 years). The mean ± SD age of all patients was 14.4 ± 2.05 years. The sex of the patients was equally distributed (45.3% male; 47.2% female; 7.5% not reported).
Furthermore, 83.0% (44 patients) of the patients had no specific medical history. In the remaining 17% of the patients, medical history such as AR (9.4%), allergy (3.8%), allergic bronchitis (1.9%) and allergic conjunctivitis (1.9%) was reported. Furthermore, 64.2% (34 patients) took no concomitant medication whereas 19 patients took antihistamines (such as loratadine (9.4%), desloratadine (7.5%), fexofenadine (5.7%), cetirizine (5.7%) and other concomitant medications (7.5%). The severity of AR at baseline was considered as mild in 10 patients (18.9%), moderate in 26 patients (49.1%) and severe in 12 patients (22.6%). For 9.4% of the patients (n=5) the severity of symptoms was not reported. In most of the patients, the treatment duration was one week (62.3%, n=33). For the other patients, the treatment duration was either two weeks (1.9%, n=1), one month (22.7%, n=12) or only reported at the beginning of the study (13.2%, n=7). For one patient (1.9%) no treatment duration was reported. The dosage of Ze 339 was in the most cases one tablet twice daily (86.8%, n=46). Some of the patients also received a dosage of one tablet three times daily (9.4%, n=5). For two patients (3.8%) the dosage was not reported.
Improvement of symptoms
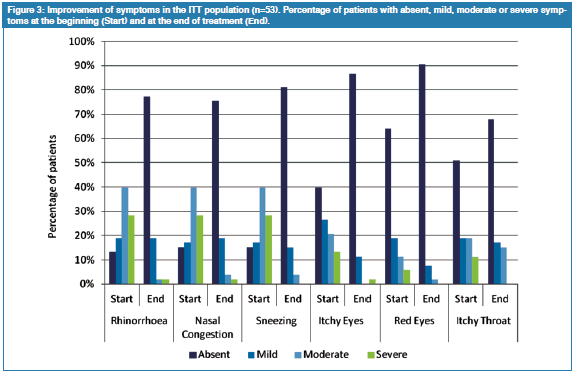
An improvement of symptoms was reported for most of the patients (83.0%, n=44) already after one week. Further 7.5% of the patients (n=4) had a time to improvement of one month. In 7.5% of the patients (n=4) the time to improvement was not reported. For one patient (1.9%) an improvement of symptoms at the beginning of the study was reported. In most of the patients the extent of improvement in symptoms of AR was reported as full recovery (52.8%, n=28) or significant improvement (34.0%, n=18, Figure 2). Most of the patients (67.9%- 90.6%) reported the symptoms to be absent at the end of treatment (Figure 3). This is also reflected by a statistically significant improvement of the single symptoms of AR (rhinorrhoea, p<0.001; nasal congestion, p<0.001; sneezing, p<0.001; itchy eyes, p<0.001; red eyes, p<0.005; itchy throat, p<0.05) at the end of treatment. The TSS and the composite scores TNSS and TNOSS (Figure 4) also decreased with statistical significance until the end of treatment (Table 1).
Concomitant medication
In the ITT population (n=53) some patients also took comedication as described above. In both groups, patients with (n=19) and without (n=34) comedication, all single symptoms of AR were improved at the end of treatment (Figure 5). The TSS, TNSS, TNOSS and the single symptoms rhinorrhoea, nasal congestion, sneezing, itchy eyes and red eyes were significantly improved in both groups (Table 1). In patients without comedication also the symptom itchy throat significantly improved, whereas the improvement in patients with comedication was not significant (Table 1). At the beginning of the treatment the TSS showed a tendency to be higher in patients with concomitant medication (p=0.082). However, all symptoms except rhinorrhoea were not statistically different between the groups. For the symptom rhinorrhoea patients with comedication had a significantly higher score at the beginning of the treatment (p=0.004) and also the change from baseline of rhinorrhoea was significantly different between patients with and without comedication (Table 1). At the end of treatment the symptom score for rhinorrhoea was similar in both groups (p=0.247).
Overall effectiveness and tolerability
The treatment was evaluated by the physicians and the patients as effective in 90.6% and 84.9%, respectively (Figure 6 a and b). Furthermore, 90.6% of the physicians and 86.8% of the patients considered the treatment as well tolerated (Figure 7 a) and b).
Discussion
In Western Europe the prevalence of AR ranges from 17-29% in adults and is frequently undiagnosed14. However, AR is already a common paediatric condition. In a worldwide study of over one million adolescents aging between 13 and 14 years, the prevalence was 14.6%15. It is not only the most common allergic disease, but it is among the top ten reasons for medical consultation16, which represents a great economic impact and decreases quality of life.
This has promoted advances in treatment and design of new strategies based on new pathogenic concepts and the impact of the patients quality of life. Although antihistamines are not a cure for AR, its use for the treatment of the condition is widespread. Antihistamines do not stop the allergic reaction and are not very effective in relieving nasal congestion, which responds better to leukotriene inhibitors and corticosteroids. Antihistamines temporarily relieve some of the symptoms of AR. However, there are possible side effects of antihistamines, which include: dry mouth, throat and eyes, and drowsiness.
For, Ze 339 two randomised controlled clinical studies confirmed the efficacy of Ze 339 in comparison with antihistamines. The effects of Ze 339 were similar to those of cetirizine or fexofenadine in patients with seasonal AR17,18. Therefore, Ze 339 is effective in relieving symptoms of AR and well tolerated. Regarding the mode of action, it is noted that a doubleblind, randomised, cross-over study investigated the efficacy and mode of action of Petasites hybridus leaf extract Ze 339, desloratadine and placebo in symptoms of AR by measuring the nasal airflow after unilateral nasal allergen provocation. An interesting finding was that Ze 339 significantly reduced IL-8 and leukotriene B4 levels. Furthermore, Ze 339 showed greater efficacy in relieving nasal obstruction and inhibition of mediators than desloratadine. Thus, the authors concluded that Ze 339 expands the therapeutic options for the symptomatic treatment of AR3.
In comparison to randomised controlled clinical studies observational studies have some limitations. Observational studies are usually performed in the relevant target population. Therefore, they possess a high external validity, however, at the potential cost of a compromised internal validity. Despite the important differences between both study types observational studies may provide complementary data for example for special populations. The present sub-analysis of a paediatric population shows that the treatment with Ze 339 was effective in the majority of paediatric patients, being considered as excellent, very good or good in 90.6% of cases evaluated by the physician and in 84.9% of cases evaluated by the patient. Furthermore, also the tolerability was evaluated as excellent, very good or good by 90.6% of the physicians and 86.8% of the patients. This response to the treatment was in accordance with the results of the total study population12.
For the evaluation of symptoms not only the nasal symptoms rhinorrhoea, sneezing and nasal congestion but also the symptoms itchy eyes and itchy throat were scored. The patients treated only with Ze 339 without comedication showed a statistically significant improvement in all single symptom scores and the composite scores TNSS and TNOSS (Table 1). In patients treated with Ze 339 and comedication the symptom itchy throat was not statistically significantly improved. This could be explained by a more severe grade of AR in these patients. At the beginning of the treatment, the symptom score for rhinorrhoea was significantly higher in patients treated with comedication than in patients treated only with Ze 339. Oral H1-antihistamines are effective in the treatment of nasal symptoms like rhinorrhoea but less of nasal congestion. For this reason, the high value of rhinorrhoea at the beginning of the treatment justified the concomitant use of antihistamines.
Also noteworthy is the fact that a rapid response to the treatment was obtained. For more than half of the patients (62.3%) the treatment duration was one week and most of the patients (86.8%) took one tablet of Ze 339 twice daily. In the summary of product characteristics, a dosage of two tablets daily is recommended for basic treatment. Therefore, our results confirm the effectiveness of the recommended dosage. This is also supported by two post-marketing surveillance studies conducted in Switzerland10,11.
The opinion of physicians and patients regarding the effectiveness as well as the tolerability of Ze 339 was comparable. This is very important due to the impact that AR has on the patients quality of life. Importantly, the results of the paediatric population greatly resemble those obtained in the general population of the original study12.
These findings, together with the fact of absence of significant adverse reaction reports allows us to suggest that Ze 339 is an effective and well tolerated therapy for the treatment of AR in children and adolescents. Ze 339 does not act like antihistamines but due to its dual mode of action comprises benefits in the treatment of the early and late allergic response in AR. Therefore, it is not only eligible for becoming a first line therapy but also an alternative for those patients who cannot be treated with antihistamines for any reason.
Conclusion
Ze 339 is effective, well tolerated and safe in the relief of all symptoms of AR. Beneficial effects were reported by patients and physicians. Ze 339 may be considered for the treatment of AR in paediatric populations.
Acknowledgements
We would like to thank Professor Jürgen Drewe and Dr. Sabine Nebel for reviewing the manuscript. We would also like to thank all physicians who took part in the observational study. For the full list of attending physicians please be referred to the publication of the original study12.
Conflict of interests
Mercedes Rodríguez de Marquis, Maria González Yibirín and Jairo J. Ruiz Rodríguez are employees of Laboratorios Leti, S.A.V., Guarenas, Venezuela. Cordula Moll is an employee of Max Zeller Söhne AG, Romanshorn, Switzerland.
Referencias
1. Bousquet J, Khaltaev N, Cruz AA, et al. Allergic Rhinitis and its Impact on Asthma (ARIA) 2008 update (in collaboration with the World Health Organization, GA(2)LEN and AllerGen). Allergy. Apr 2008;63 Suppl 86:8-160. [ Links ]
2. Schapowal A. Treating intermittent allergic rhinitis: a prospective, randomized, placebo and antihistamine-controlled study of Butterbur extract Ze 339. Phytother Res. May. 2005;19(6):530-537. [ Links ]
3. Dumitru AF, Shamji M, Wagenmann M, et al. Petasol butenoate complex (Ze 339) relieves allergic rhinitis-induced nasal obstruction more effectively than desloratadine. J Allergy Clin Immunol. Jun 2011;127(6):1515-1521. [ Links ]
4. Thomet OA, Schapowal A, Heinisch IV, Wiesmann UN, Simon HU. Anti-inflammatory activity of an extract of Petasites hybridus in allergic rhinitis. Int Immunopharmacol. Jun 2002;2(7):997-1006. [ Links ]
5. Schapowal A. Butterbur Ze339 for the treatment of intermittent allergic rhinitis: dose-dependent efficacy in a prospective, randomized, double-blind, placebo-controlled study. Arch Otolaryngol Head Neck Surg. Dec 2004;130(12):1381-1386. [ Links ]
6. Thomet OA, Wiesmann UN, Blaser K, Simon HU. Differential inhibition of inflammatory effector functions by petasin, isopetasin and neopetasin in human eosinophils. Clin Exp Allergy. Aug 2001;31(8):1310-1320. [ Links ]
7. Thomet OA, Wiesmann UN, Schapowal A, Bizer C, Simon HU. Role of petasin in the potential anti-inflammatory activity of a plant extract of petasites hybridus. Biochem Pharmacol. Apr 15 2001;61(8):1041- 1047. [ Links ]
8. Berger D, Burkard W, Schaffner W. Influence of Petasites hybridus on dopamine-D2 and histamine-H1 receptors. Pharm Acta Helv. Feb 1998;72(6):373-375. [ Links ]
9. Scheidegger C, Dahinden C, Wiesmann U. Effects of extracts and of individual components from Petasites on prostaglandin synthesis in cultured skin fibroblasts and on leucotriene synthesis in isolated human peripheral leucocytes. Pharm Acta Helv. 1998;72(6):376-378. [ Links ]
10. Keusch A, Notter D, Brattstrom A, Morandell D, Polasek W. Treatment of the allergic rhinitis with the Butterbur special extract Ze 339. Ars Medici. 2004;10:509-515. [ Links ]
11. Käufeler R, Polasek W, Brattström A, Koetter U. Efficacy and safety of butterbur herbal extract Ze 339 in seasonal allergic rhinitis: postmarketing surveillance study. Adv Ther. Mar-Apr 2006;23(2):373-384. [ Links ]
12. Rodríguez de Marquis M, González Yibirín M. Post marketing report: effectiveness of the standardized extract of leaves of Petasites hybridus (Tesalin®) in the treatment of allergic rhinitis. AVFT 2012;31(1):11-16. [ Links ]
13. FDA. Guidance for Industry: Allergic Rhinitis: Clinical Development Programs for Drug Products. Draft Guidance. 2000. [ Links ]
14. Bauchau V, Durham SR. Prevalence and rate of diagnosis of allergic rhinitis in Europe. The European respiratory journal. Nov 2004;24(5):758-764. [ Links ]
15. Aït-Khaled N, Pearce N, Anderson HR, Ellwood P, Montefort S, Shah J. Global map of the prevalence of symptoms of rhinoconjunctivitis in children: The International Study of Asthma and Allergies in Childhood (ISAAC) Phase Three. Allergy. Jan 2009;64(1):123-148. [ Links ]
16. Gregory C, Cifaldi M, Tanner LA. Targeted intervention programs: creating a customized practice model to improve the treatment of allergic rhinitis in a managed care population. The American journal of managed care. Apr 1999;5(4):485-496. [ Links ]
17. Schapowal A. Treating Intermittent Allergic Rhinitis: A Prospective, Randomized, Placebo and Antihistamine-controlled Study of Butterbur Extract Ze 339. Phytother Res. Jun 2005;19(6):530-537. [ Links ]
18. Schapowal A. Randomised controlled trial of butterbur and cetirizine for treating seasonal allergic rhinitis. BMJ. Jan 19 2002;324(7330):144-146. [ Links ]