Introduction
Creatine is a biochemical compound synthesized from the amino acids arginine, glycine, and methionine, primarily in the liver, kidneys, and pancreas. Its primary function is to store and provide energy in the form of phosphocreatine (PCr), especially in tissues with high energy demands, such as skeletal muscles and the brain. PCr acts as a phosphate donor to regenerate ATP (adenosine triphosphate), which is the primary source of cellular energy during intense, short-duration physical activities 1. More than 95% of Cr in the human body exists in skeletal muscle, while the remaining 5% is distributed in the brain and testicles. Approximately two-thirds of muscular Cr is found to be PCr, while the remaining third is free. About 1-2% of this muscle Cr is broken down as creatinine and excreted in the urine 2.
Cr reserves in the human body are proportional to muscle mass, and adults consume approximately 1 to 3 grams daily in normal conditions. About half of the requirements are obtained from the diet, mainly from red meat and shellfish 3. The remaining half is synthesized in the liver and kidneys from the amino acids arginine and glycine by the action of the enzyme arginine glycine aminidinotransferase (AGAT) that transforms them into guanidinoacetate (GAA) that is subsequently methylated by guanidinoacetate N-methyltransferase (GAMT) using S-adenosyl methionine to form creatinine 4.
Cr is a popular ergogenic aid among athletes and has been consistently shown to increase muscle availability of Cr and PCr, improving athletes' capacity for acute exercise, as well as training adaptations, allowing performance with greater quality, favoring a better post- exercise recovery, preventing injuries, benefiting thermoregulation, rehabilitation and providing both spinal and cerebral neuroprotection 5,6,7.
In addition, given its effects on cellular energy balance regulation and reduction of oxidative stress, clinical applications have been attributed to it, finding beneficial effects after its supplementation in neurodegenerative diseases such as muscular dystrophy 8, Huntington's disease 9, Parkinson's disease 10, diabetes 11, osteoarthritis 12, fibromyalgia 13, cerebral or cardiac ischemia 14, depression 15, and aging positively influencing cognitive function 16. Some studies have even found beneficial effects in pregnant women, improving the fetus’s growth, development, and health 17.
The Cr supplementation was evaluated in short and long-term effects, as well as among healthy or patients with a particular pathology; Cr supplementation doses range between 0.3 and 0.8 g/kg/day for a maximum of 5 years, with good tolerance, without reporting significant adverse effects. The only consistently reported side effect is weight gain 18.
Cr is an effective and safe ergogenic supplement for adults and older people, but the evidence from the pediatric age group has not been conclusive. Cr has been studied in the pediatric population, particularly in various diseases affecting muscle function and cellular energy. Its use as an ergogenic supplement has been explored in conditions such as muscular dystrophy, metabolic diseases, and neuromuscular disorders. Research has demonstrated that Cr supplementation may offer potential benefits in improving muscle strength, physical function, and quality of life in children and adolescents with these conditions. Additionally, it has been suggested that Cr could play a neuroprotective role, making it an interesting option for the treatment of neurological disorders in childhood 19.
Recently, Jagim & Kerksick 20 and Metzger et al. 21 grouped the relevant and available information related to Cr supplementation in children and adolescents. However, the results are inconclusive and widespread. Therefore, this review aims to analyze the impact of different doses of Cr on physical performance and muscle function in children and adolescents in various clinical scenarios.
Materials and methods
Study design
This systematic review is aimed under the Preferred Reporting Items for Systematic Reviews (PRISMA) 22. The study population includes all reports about Cr supplementation during childhood and adolescence between 1997 and May 2023 in the electronic database search.
Selection criteria
The methodological quality assessment where the selection criteria are specified, and the risk of bias 23 are described in Tables 1 and 2, respectively. The intervention, control, and outcome parameters were selected following the Population, Interventions, Control, and Outcome (PICO) format 24:
Population: Children and adolescents aged 2 to 18 years old with Cr supplementation.
Intervention: Oral Cr supplementation.
Control: Placebo.
Outcome: Muscular function and physical performance
Research question: How does oral Cr supplementation affect muscle function and physical performance in children and adolescents?
Table 1 Methodological quality assessment.
| Step | Description |
|---|---|
| 1. Protocol development | Define and register objectives, such as evaluating the effectiveness of creatine supplementation in children and adolescents. |
| 2. Literature search | Conduct searches in relevant databases (PubMed and Google scholar) using terms like “Creatine supplementation AND children”, “Creatine supplementation AND teenagers”, “Creatine supplementation AND young athletes”, “Creatine Supplementation and adolescent athletes”, “Creatine AND Muscular function” and “Creatine AND Physical performance”. |
| 3. Study selection | Apply inclusion criteria (clinical trials Cr supplementation on children and adolescents, function and physical performance measurements) and exclusion criteria (unreported data, case reports, and pilot studies). |
| 4. Data extraction | Collect data on dosage, duration of supplementation, and outcomes related to strength and physical performance. |
| 5. Quality assessment | Assess the quality and risk of bias using the Cochrane Risk of Bias Tool. |
| 6. Analysis and synthesis | Perform qualitative analysis of the data on the effects of creatine on strength and performance. |
| 7. Reporting results | Present results clearly, including a flow diagram of study selection and summary tables. |
| 8. Discussion | Discuss findings about previous studies, highlighting the benefits and limitations of creatine in the pediatric population. |
| 9. Conclusions and recommendations | Provide conclusions on the safety and effectiveness of creatine in children and adolescents and suggest areas for future research. |
| 10. Transparency and reproducibility | Document all processes, ensuring methods are reproducible by other researchers. |
Table 2 Risk of bias
| Study | Randomization | Allocation Concealment | Blinding of Participants and Personnel | Blinding of Assessors | Incomplete Data | Selective Reporting | Other Bias |
|---|---|---|---|---|---|---|---|
| 1997_Grindstaff 25 | Low | Low | Low | Unclear | Low | Low | Unclear |
| 2002_Dawson 26 | Low | Low | High | Unclear | Low | Low | Unclear |
| 2003_Louis 8 | Unclear | Low | Low | Unclear | Low | Low | Unclear |
| 2004_Ostojic 27 | Unclear | Low | Low | Unclear | Low | Low | Unclear |
| 2004_Tranopolsky 28 | Low | Low | Low | Unclear | High | Low | Unclear |
| 2005_Escolar 29 | Low | Low | Low | Unclear | Low | Low | Unclear |
| 2007_Silva 30 | Low | Low | Low | Unclear | Low | Low | Unclear |
| 2007_Wong 31 | Low | Low | Low | Unclear | Low | Low | Unclear |
| 2009_Juhász 32 | Low | Low | Low | Unclear | High | Low | Unclear |
| 2010_Banerjee 33 | Low | Low | High | Unclear | Low | Low | Unclear |
| 2012_Mohebbi 34 | Unclear | Unclear | Low | Unclear | Low | Low | Unclear |
| 2013_Deminice 35 | Low | Low | Low | Unclear | Low | Low | Unclear |
| 2014_Hayashi 36 | Low | Low | Low | Unclear | Low | Low | Unclear |
| 2016_Solis 37 | Low | Low | Low | Unclear | Low | Low | Unclear |
| 2017_Wang 38 | Low | Low | Low | Unclear | High | Low | Unclear |
| 2017_Yañez-Silva 39 | Low | Low | Low | Unclear | High | Low | Unclear |
| 2018_Juhász 40 | Low | Low | Low | Unclear | High | Low | Unclear |
| 2019_Da Silva 41 | Low | Low | Low | Unclear | High | Low | Unclear |
| 2021_Dover 42 | Low | Low | Low | Unclear | High | Low | Unclear |
| 2022_Vargas-Molina 43 | Low | Low | High | Unclear | Low | Low | Unclear |
Search strategy and data extraction
The electronic data search of relevant references was performed in electronic databases without publication date restrictions. The electronic databases used were PubMed and Google Scholar. All reviewers retrieved selected studies as full-text reports to be rescreened in detail to confirm whether the studies met the inclusion criteria.
Variables: Because of the heterogeneity of the previous reports and the variety of variables measured in the studies, the variables were classified into two groups:
Muscular function: Hand grip, total manual muscle, quantitative muscle testing, leg press, bench press, segmental lean mass, ankle plantar flexion peak torque, right-hand grip, left-hand grip, manual muscle testing (MMT), and quantitative measure muscle strength (QMT).
Physical performance: Heat, power sprint, swimming, anaerobic work, maximal voluntary contraction (MVC), time to exhaustion, vertical jump (VJ), power test (PT), shuttle run, dribble, time to walk, time to climb stairs, swimming velocity (MSV25), active drag force (Df), hydrodynamic coefficient (Cx), power output (Po), lower limb functional grade, mechanical power output, maximum power, minimum power, fatigue index, time stands, time up-and-go, post- activation potentiation (PAP), pain intensity, rate of perceived exertion (Borg´s Scale), squat jump (SJ), drop jump (DJ), countermovement jump (CMJ), Abalakov jump (ABJ), peak work, moderate vigorous physical activity, vigorous physical activity, max jump, Wingate Anaerobic Test (WAnT), peak power output (PPO), mean power output (MPO), total work, time to stand, time to climb stairs, time to run, repeated sprint test, accuracy of shooting and, dribble.
Reported data finding
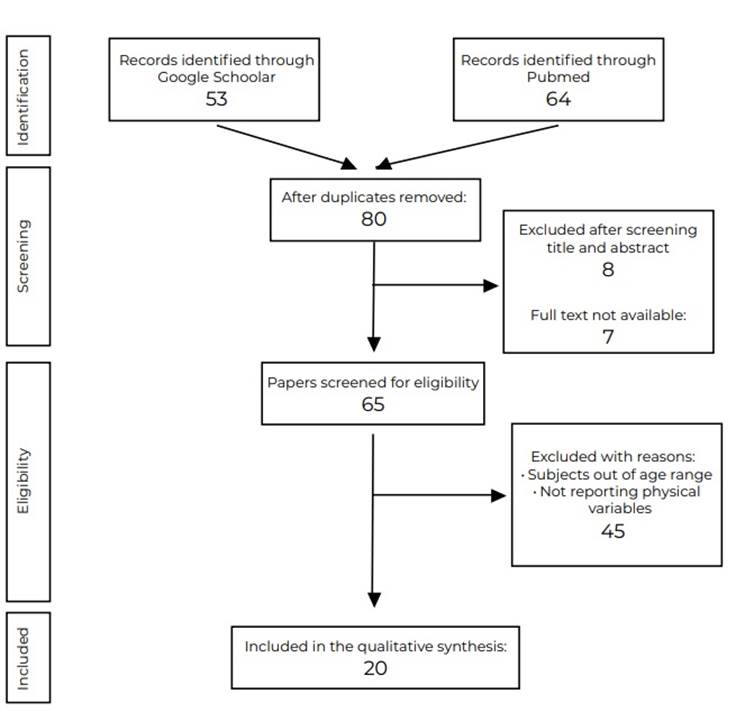
The electronic search of databases included 117 potential studies, as shown in Figure 1. After removing duplicates (n=37), the reports with different study designs in the abstract (n= 8) were discarded; after the full-text available studies (n=65), the manuscripts were screened in more detail and assessed to comply with the inclusion criteria, the manuscripts removed after that whit reasons where: age range (n=35) and reporting physical variables (n= 10). Finally, the remaining 20 studies were included 8,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43.
Characteristics of included studies
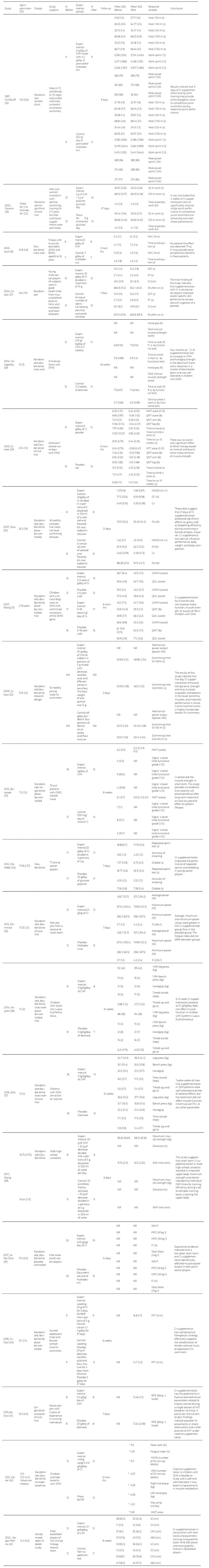
The included studies are listed in Table 3. All studies were Randomized Clinical Trials (RCTs), 14 double-blinded, three single- blinded, and two open-labeled; 5 were crossover, and 15 were parallel. All studies reported were in English from 1997 to May 2022. The supplementation periods were from 5 days to 6 months, with doses of 0.03, 0.1, 0.15, 0.3, 2.0, 3.0, 5.0, 10.0, 20.0, 21.0, and 30gr/day. The principal outcome measures were physical variables divided into two groups: Muscular function using force measures and physical performance using time, distance, and power measures.
Tabla 3 Characteristics of Included Studies in Qualitative Synthesis
SD: standard deviation; g: grams; Cr: creatine; CrM: creatine monohydrated; s: second; J: Joule; W: Watt; kJ: kilojoule; m: meter; DMD: Duchenne muscular dystrophy; BMD: Becker muscular dystrophy; MVC: maximal voluntary contraction; N·m: Newton·meter; SDT: specific dribble test; PT: power test; VJ: vertical jump; cm: centimeter; kg: kilogram; NR: not reported; N: Newton; ft: feet; FFM: fat-free mass; MMT: manual muscle testing; QMT: quantitative muscle testing; lb: pound; ml: milliliters; MSV25: 25 m swimming velocity; m.s-1: meter per second; Df: active drag force; Cx: hydrodynamic coefficient; Po: power output; SMA: spinal muscular atrophy; GMFM: gross motor function measure; QOL: parent questionnaire for the PedsQL Neurouscular Module; kW: kilowatt; mg: milligrams; CI: confidence interval; W/s-1: Watt/second; 1-RM test: one-maximum repetition test; reps: repetitions; JDM: juvenile dermatomyositis; PCr: phosphocreatine; PAP: post-activation potentiation; min: minute; WAnT: Wingate anaerobic test; PPO: peak power output; W.kg-1:Watts per kilogram of body weight; MPO: mean power output; FI: fatigue index; J.kg-1: Joules per kilogram of body weight; PFT: plantar flexion peak torque; RPE: rate of perceived exertion; HIIT: high intensity interval training; MVPA: moderate-vigorous physical activity; VPA: vigorous physical activity; *: Mean difference between experimental and placebo; SJ: squad jump; DJ: drop jump; CMJ: counter movement jump; ABJ: Abalakov jump.
Results
Effect of 0.03 g/kg/day of monohydrate creatine
A study with this dose compared the Cr supplementation and maltodextrin as the placebo for 14 days; this was a parallel design. Yañez-Silva et al. (35) found that Cr supplementation benefited muscle power output in elite youth soccer players and suggested that the dose used appears to be the lowest effective dose of Cr seen in the literature. They found significant changes in the two groups.
Effect of 0.1-0.15 g/kg/day of monohydrate creatine
Five studies evaluated the effect of Cr supplementation in adolescents 28,36,37,42,43 using variables like muscle function, muscle strength, and aerobic capacity parameters. Three of these were cross-over trials 28,36,43 and two studies were parallel designs 37,42. Four studies used a dose of 0.1g /kg/day 28,36,37,43 another one used 0.15g/kg/day 42, and the placebo was dextrose. Solis et al. 37 and Hayashi et al. (36) Solis et al. evaluated the effect of Cr supplementation on muscular function in 12 weeks in different diseases, 37 found that Cr supplementation did not affect muscle function in juvenile dermatomyositis (JDM) Hayashi et al. 36 reported no significant changes between placebo and Cr for any muscle function in systemic lupus erythematosus. However, Tarnopolsky et al. 28 reported that four months of Cr supplementation does not improve functional tasks or activities of daily living like walking or climbing stairs in Duchenne muscular dystrophy (DMD). Vargas-Molina et al. 43 reported that eight weeks Cr supplementation with resistance and plyometric training increased the lower-limb Abalakov jump power and scoring performance in under 16 years of basketball players. Dover et al. 42 used a dose of 0.15 g/kg/day and reported that Cr supplementation in children with JDM did not affect muscle function, strength, and aerobic capacity. In summary, the evidence has shown that Cr supplementation with a dose of 0.1g/kg/day only affected muscular strength in conjunction with resistance training.
Effect of 0.3 g/kg/day of monohydrate creatine
Two studies with this dose versus placebo 35,41 were evaluated. One of the studies was single-blind cross- over design 41, the other was a randomized, double- blind design. The studies evaluated pre- and post- measure with a supplementation time of 7 days, using dextrose and maltodextrin as placebo. The subjects were soccer players aged 15 to 19. Da Silva Azevedo et al. 41 evaluated the effect of Cr in 8 males measured by the rate of perceived exertion (with Borg´s scale). The data suggest a possible improvement in shock attenuation and a safer practice of high-intensity interval training (HIIT) under Cr supplementation. On the other hand, Deminice et al. 35 they measured the Cr in 25 young high-performance athletes to determine its effect after intense exercise sessions. They analyzed different muscular and metabolic parameters, finding that Cr supplementation favored more excellent muscular function. In summary, supplementing 0.3g/day of Cr for 7 days in young high-performance athletes allows better impact control and excellent discharge attenuation to carry out high-intensity training sessions more safely.
Effect of 2-3 g/day of monohydrate creatine
Cr supplementation was analyzed in children with muscle disease. Wong et al. 31 studied patients with spinal muscular atrophy (SMA) aged 2-18 years in a double-blind randomized clinical trial in parallel, administering 2g/day of Cr vs. placebo, in which this dose was only administered when the children were under 5 years for 6 months, the muscular function were evaluated at different times without finding significant effects, concluding with this that under experimental conditions the supplementation of 2g/day of Cr for 6 months in children with spinal muscular atrophy does not improve motor function and muscle strength of these patients. However, Louis et al. 8, in a double-blind cross-over study carried out on children with muscular dystrophy, who were administered 3 g/day of Cr for 3 months compared to placebo, this administration improved muscle strength and resistance to fatigue.
Effect of 5 g/day of monohydrate creatine
Four studies evaluated the effect of Cr versus placebo 26,29,31,33, two studies had a single-blinded design (26,33), and the other was double- blinded (29,31). All studies evaluated a pre and post- measure with a different supplementation time, and only two studies described the placebo group glucose polymer powder 26, and vitamin C 33. Dawson et al. 26 evaluated the effect of Cr on male swimmers (16.1 ± 1.4 years) and females (16.6 ± 2.2 years) for 27 days with a maximum speed test of 50 meters (m) and 100 m freestyle, in which no significant differences were found between the Cr and placebo groups, while in the total anaerobic work output score (kilojoule) the Cr group increased their scores after supplementation. Other studies evaluated the effect of Cr on different pathologies, such as those by Wong et al. 31 the study included children with SMA (5 to 18 years) and determined that six months of Cr supplementation at this dose did not improve motor function or muscle strength. The remaining studies assessed children with DMD, in the case of Banerjee et al. 33 performed an 8-week supplementation and reported a significant difference in MMT score between the Cr and placebo group after the supplementation. On the other hand, Escolar et al. 29 supplemented for six months and didn’t find a statistically significant difference in the MMT or the QMT scores. In summary, the findings reported by these authors suggest that a 5 gr Cr supplementation effectively increases the scores of total anaerobic works in swimmers, but the effect on different pathologies is not yet apparent.
Six articles evaluated Cr supplementation versus placebo in adolescents 25,30,32,34,38,40 six articles were double-blind. All studies assessed pre and post- measures with different supplementation times and placebo substances (see Table 1). The subjects range in age from 12 to 19 years. Silva et al. 26 evaluated the effect of Cr in 16 females for 21 days, where Df, Cx, Po, and swimming velocity (MSV25) were assessed. The data suggest that Cr supplementation significantly affected gross and/or propelling efficiency during swimming in female athletes. On the other side, Juhász, et al. 32 evaluated 16 young men for five days, where swimming time over 100 meters (1st and 2nd) were assessed, having as results that Cr supplementation enhances the dynamic strength and may increase anaerobic metabolism in the lower extremity muscles. Again, Juhász et al. 40 measured 18 male and female competitive swimmers with tendon injuries. The data suggest that after six weeks of Cr supplementation and rehabilitation, it effectively supports the rehabilitation of tendon overuse injuries in adolescent fin swimmers. Wang et al. 38 identified in 17 male high school canoeists, after six days of supplementation with Cr, that maximum muscle strength and the neuromuscular phenomenon of PAP improves the efficiency during a set of complex training bouts involving the upper body. Mohebbi et al. 34 rated 17 young soccer players and concluded that repeated sprints and dribbling performance improved after seven days of supplementation with Cr. Finally, Grindstaff et al. 25, evaluated 18 males and females for nine days where heat to 50 and 100 m (three times each), work sprint (three times), and peak power sprint (three times) were measured, where the results indicated that supplementation during swim training may provide some ergogenic value in competitive junior swimmers during repetitive sprint performance. In summary, the evidence shows that Cr supplementation with a dose of 20-21g/day produced significant and positive efficiency effects during swimming and in soccer skills on male and female young athletes.
Effect of 30 g/day of monohydrate creatine
Only One study reported this dose versus placebo (cellulose) in adolescents 27, in a pre-and post-measures design. The subjects ranged in age from 14 to 19, and all were soccer players from the junior league. Ostojic et al. 27 evaluated the effects of Cr supplementation in 55 males and females for seven days, where dribble, sprint power, endurance, vertical jump, and shuttle run. The author indicates that supplementation in young soccer players improved soccer- specific skills but not endurance performance.
Discussion
Cr is a popular ergogenic supplement among athletes due to its beneficial effects on muscle and energy metabolism. Likewise, it has also been helpful in some neurodegenerative diseases in the adult population. This systematic review compiles the studies carried out in children and adolescents, demonstrating their benefits in different clinical settings.
The Cr supplementation in athletes from different disciplines like swimmers 25,26,30,32,40, canoeists 38, soccer 27,34,35,39,41, and basketball players 43 improves most of the physical performance and muscle function. This could be explained by the role of the creatine kinase (CK)/PCr system, as Wallimann et al. suggest 44. Since the primary metabolic function of Cr is to form PCr from the union with a phosphoryl group (Pi) by the CK enzyme, the Pi released in the degradation of Adenosine triphosphate (ATP) to adenosine diphosphate (ADP) provides energy for metabolic activity, together can use in the resynthesize of ATP and help buffer energy needs, which explains the potential ergogenic and therapeutic benefits of Cr supplementation increasing the availability of muscle Cr.
This suggests that some physical activity is necessary to take advantage of the Cr reserves generated by supplementation, concurring with Harris et al. 45 who evaluated the interaction between exercise and Cr supplementation in subjects who performed 1 hour of bicycle ergometer exercise daily with only one leg, while the other leg served as a control. It was shown that there is an increase in Cr muscle reserve when supplementation is combined with regular exercise. However, in the most recent review in this field, Metzger et al. 21 concluded that more well- conducted trials are needed to determine whether Cr supplementation is effective in young athletes.
The dose of Cr is essential to the observed variables because some authors found the best results at specific doses. Vargas et al. 43, administrated a 0.1gr/kg/day dose. They evaluated basketball players undergoing a resistance and plyometric training program and suggested that de Cr supplementation might benefit strength-training adaptations and sports performance. Mills et al. 46 found similar effects because they evaluated physically active young adults during training sessions, finding that this is an effective strategy for increasing muscle strength and endurance. However, Pakulak et al. 47 evaluate young adults, and they did not find the effects of Cr supplementation on muscle strength and endurance. They only report that Cr supplementation and resistance training slightly improve knee extensor muscle accretion.
When the Cr supplementation was evaluated in patients with an associated pathology, the reports did not demonstrate significant improvements, for example, in patients with DMD 8,28,29,33, SMA 31, juvenile dermatomyositis 37,42, or lupus erythematosus 36. Additionally, the protocols were very heterogeneous, and the doses differed between studies. Banerjee et al. specifically reported Cr as a treatment for some diseases, like DMD, to alleviate the symptoms of muscular dystrophy 33. They evaluated children with DMD who were administered 5 gr/day of Cr for eight weeks and concluded that Cr improved the cellular energetics of their calf muscle with the preservation of muscle strength. However, they suggest no evidence of a beneficial effect after long-term treatment, concurring with Walter et al. 48, which showed little to no benefit of Cr supplementation in patients with myotonic and muscular dystrophies. Still, their patients were adults and not only with DMD, but they also included sarcoglycan-deficient limb-girdle muscular dystrophy and Beker dystrophy, while Tarnopolsky et al. 28 It was reported that Cr supplementation in children at a dose of 0.1 gr/kg/day for four months increased fat-free mass and handgrip strength in the dominant hand. Hence, Cr supplementation has a beneficial effect on DMD after long-term treatment.
One of the biggest problems in Cr supplementation is the dose and frequency. Each study uses a different administration protocol in different subject contexts, which makes summarizing the evidence difficult. Additionally, the response variables are very heterogeneous regarding evaluation method and timing. The authors must consider the CONSORT guidelines 45 to improve the quality of the trials and answer whether Cr applies to children and adolescents.
Some researchers suggest that higher doses of Cr may yield more pronounced effects, particularly in enhancing athletic performance and muscle recovery. However, this potential benefit must be weighed against the possibility of increased adverse effects, emphasizing the urgent need to establish clear and comprehensive guidelines for supplementation. Additionally, the current literature reveals a notable gap in long-term studies investigating the impact of Cr on children's physical and cognitive development, which raises significant concerns among healthcare professionals regarding its safety and efficacy 19.
The studies included in this review exhibit considerable variability in their methodologies, populations, and outcome measures. This heterogeneity complicates the ability to draw definitive conclusions and limits the generalizability of the findings across different groups. Additionally, the quality of the included studies varies; some lack rigorous design, control groups, or adequate randomization. Most studies focus on specific populations, such as athletes or individuals with particular health conditions, which may not represent the broader population. Consequently, this specificity limits the applicability of the findings to other groups.
Conclusions
Current evidence leaves the effects of Cr supplementation in children and adolescents uncertain. Most studies indicate a positive impact on physical performance, particularly among young athletes, with higher doses showing more significant benefits during physical activity. However, the dosages, administration regimens, and protocols vary widely across studies.
Establishing optimal protocols is crucial to determining effective and safe dosages for the pediatric population, considering age, weight, physical activity level, and existing health conditions. Advancing these research areas could lead to a better understanding of the effects and benefits of Cr in children and adolescents and the development of evidence-based guidelines for its use