INTRODUCTION
Dental pulp, situated at the innermost tissue within teeth, primarily comprises loose connective tissue and plays an essential role in the repair and regeneration of dental tissues. Traumatic incidents or the impact of infectious agents and other pathogenic stimuli can readily give rise to pulpitis 1. Human dental pulp cells (hDPCs) are present within dental pulp tissue, constituting a mixture of fibroblasts, inflammatory, immune cells, odontoblasts, and undifferentiated mesenchymal cells 2,3. Among these components, the undifferentiated mesenchymal cells possess a multipotent cellular phenotype with the potential to differentiate into various cell lineages, such as osteoblasts, adipocytes, neural progenitors, and chondrocytes 4-6. When dental pulp tissue is exposed to external stimuli, dental pulp cells can differentiate into odontoblasts under the regulation of various factors, which is a significant factor in the regeneration and repair capacity of dental tissue damage7,8. Therefore, exploring the mechanisms of hDPCs proliferation and odontoblastic differentiation is essential.
Cell-based tissue engineering has become an irreplaceable method for regenerating dental tissues 9. Its fundamental mechanism involves inducing stem cells to differentiate into odontoblast-like or osteoblast-like cells, ultimately achieving the regeneration of the dental pulp-dentin complex 10,11. Furthermore, hDPCs exhibit some advantages, characterized by no ethical controversy, low immunogenicity, and easy obtainment from impacted or orthodontic extraction 12,13. Therefore, hDPCs have emerged as a potential alternative for future use in regeneration therapy. In addition to stem cells and biomimetic materials, suitable growth factors are imperative in tissue regeneration by promoting cell proliferation, differentiation, and locomotion 14,15.
Insulin-like growth factor-1 (IGF-1), a member of the insulin-like peptide family, exerts essential roles in the bone formation and periodontal regeneration of teeth 14,16,17. Local controlled delivery of IGF-I from dextran-co-gelatin hydrogel microspheres enhances new bone formation and alveolar bone reconstruction in the periodontal defects 17. Moreover, IGF-1 is also involved in the odontoblastic differentiation of hDPCs or human dental pulp stem cells (hDPSCs). In vitro experiments showed 100 ng/mL IGF-1 promotes proliferation and increases the osteogenic differentiation-related expression of DPSCs via mammalian target of rapamy cin (mTOR) signaling pathway 18, as well as the enhancement of proliferation and osteo/ odontogenic differentiation was found in the human periodontal ligament stem cells via activating MAPK pathways 19. Similarly, other in vitro research indicates that combined use of 100 ng/ml VEGF and 100 ng/mL IGF-1 also improve the proliferation, migration, osteogenesis, and angiogenesis of hDPSCs via activation of the phosphoinositide 3-kinase (PI3K)/Akt signaling pathway 20.
This study aimed to investigate the effect of IGF-1 on the proliferation and odontogenic differentiation of hDPCs in vitro and in vivo. These findings may offer valuable insights into the potential utility of hDPCs in treating dental pulp diseases.
MATERIALS AND METHODS
Isolation of human DPCs
Healthy premolars for orthodontic needs or third molars were collected from patients without dental caries or periodontal tissue diseases at the dental clinic of The Second Hospital of Hebei Medical University. All study protocols were approved by the Ethics Committee of The Second Hospital of Hebei Medical University, and written informed consent was obtained from all patients. The hDPCs were obtained as described previously. To completely collect the dental pulp, the tooth surface attachments were scraped off, and the teeth were split in a sterile environment. After washing three times with phosphate buffer solution (PBS), dental pulp tissues were cut into pieces of 1 mm×1 mm in size, then digested in a solution of 3mg/ mL collagenase type I (Sigma-Aldrich) and 4mg/mL dispase (Sigma-Aldrich) at 37°C on a shaker for one hour. Following filtration and centrifuging, cell suspensions of dental pulp were cultured in Dulbecco’s modified Eagle’s medium (DMEM, Gibco, Grand Island, NY, USA) supplemented with 20% fetal bovine serum and 1% penicillin-streptomycin (Gibco, Grand Island, NY, USA) at 37°C in a 5% CO2 incubator. Observe whether dental pulp cells crawl out of the tissue blocks and cell morphology and growth. Once cells adhere to the culture surface, the medium was refreshed every three days. When cell fusión reached over 80%, the adherent cells were digested with 0.25% trypsin (Sigma-Aldrich), and passage culture was performed at a 1:3 ratio. The hDPCs from the mixed population at passages 3 to 6 were used for subsequent experiments.
Immunohistochemical staining
Passage 4 hDPCs were digested with 0.25% trypsin and prepared as cell suspension, then seeded into 6-well cell culture plates at a concentration of 1×104 cells per well. Immunohistochemical staining for vimentin and cytokeratin was carried out on the day following cell adherence. Briefly, cell slides were fixed with 4% formaldehyde for 10 minutes, then treated with 0.5% Triton X-100 (diluted in PBS) for 10 minutes at room temperature. Following PBS washing, cells were blocked with 10% FBS for 20 minutes and subsequently incubated overnight at 4°C with primary antibodies anti-vimentin and anti-cytokeratin (1:200, Abcam). After washing three times with PBS, cells were incubated with biotin-conjugated secondary antibodies (1:1000, Abcam) at room temperature for 20 minutes. DAB staining was performed, and counterstaining with hematoxylin was carried out to visualize the cell nuclei. After mounting coverslips with neutral balsam, photography was conducted under an optical microscope (Olympus, Tokyo, Japan).
Flow cytometry analysis
The purity of isolated hDPCs was assessed using flow cytometry analysis with specific antibodies for CD44 and CD45 21. The cells were trypsinized and fixed in 4% paraformaldehyde in PBS. Subsequently, hDPCs were permeabilized with 0.1% Triton X-100 (Sigma-Aldrich) in PBS and then incubated with conjugated antibodies against CD44 (1:100, Biotech, Minneapolis, USA) and anti-CD45 (1:100, Biotech, Minneapolis, USA). After washing three times with PBS, flow cytometric analysis was conducted using a FACS (Becton, Dickinson, San Jose, CA) with Quest CELL software (Becton, Dickinson).
Cell proliferation assay
The proliferation ability of hDPCs was assessed using the MTT (3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyl nyltetrazolium bromide, Sigma) assays. Passage 4 hDPCs were prepared as a cell suspension with 0.25% trypsin and seeded into 96-well cell culture plates at a concentration of 2×103 cells per well. After 24 hours of cultivation, cells were randomly divided into four groups. In the experimental groups, DMEM culture medium containing different concentrations of IGF-1 (50 ng/mL, 75 ng/mL, 100 ng/mL, Peprotech, USA) was added, while in the control group, DMEM without IGF-1 was added. Each group had five replicates. The cells were then cultured at 37°C for 1, 3, 5, and 7 days, respectively. Subsequently, 20μl of MTT (2 mg/mL in PBS) was added to each well. After 4 hours of incubation, the supernatant was removed, and 150μL DMSO were added. The absorbance values at A450 for each well were determined using a microplate reader (Bio-Rad).
Odontoblastic differentiation in vivo
The acellular dermal matrix (ADM, Beijing Qingyuan Weiye Company, China) underwent aseptic cutting into pieces measuring 5mm×5mm×1mm. These pieces were subjected to three washings with PBS and a 2-hour soak in 10% FBS within a sterile environment. The 2x105 hDPCs were prepared as a cell suspension. After removing the FBS, cells were gently seeded onto the scaffold using a pipette, ensuring even distribution within the scaffold interstices through gentle shaking. The composite was co-cultured at 37°C for 24 hours. The culture medium was replaced with 100 ng/mL IGF-1 culture medium supplemented with mineralization induction solution, consisting of 10% FBS, DMEM, 10 mmol/L β-glycerophosphate, 100 mg/mL vitamin C, and 10 nmol/L dexamethasone. The culture medium in the control group was replaced with a mineralization induction solution without IGF-1. The induction culture was maintained for three days. For the in vivo phase, eight 4-week-old immunodeficient mice were selected. Under sterile conditions, incisions were made in the skin along their dorsal midlines. The hDPCs-scaffold complexes were transplanted subcutaneously into the left dorsal region, while control scaffolds were transplanted as controls into the right dorsal region. The odontoblastic differentiation in the hDPCs-scaffold complexes within the mice was observed at two and four weeks post-transplantation.
Histological staining
Hematoxylin and Eosin (HE) staining was used for the histological observation, and Alizarin red staining was performed to identify calcium-containing osteocytes or odontoblasts. The mice were euthanized at two and four weeks post-transplantation. The scaffold and surrounding tissues were immediately harvested and fixed in 4% paraformaldehyde. Subsequently, 4-μm-thick sections were prepared using a freezing microtome. The sections were stained with hematoxylin for 3 minutes for HE staining, followed by differentiation and a 20-second rinse in alkaline water. Next, sections were stained with eosin for 20 seconds. For the alizarin red staining, sections were fixed in 95% anhydrous ethanol for 10 minutes. After rinsing in double-distilled water thrice, sections were stained in 0.1% alizarin red-Tris-HCl at 37°C for 30 minutes. All sections were observed and counted under a light microscope (Olympus, Tokyo, Japan).
Transmission electron microscopy (TEM)
TEM was used to assess the morphologies of hDPCs-scaffold complexes. After two and four weeks post-transplantation, the scaffold was immediately harvested and washed three times with PBS, then fixed in 4% (v/v) glutaraldehyde for 2 hours at room temperature. The complexes were dehydrated with varying ethanol concentrations and embedded with epoxy resin. Subsequently, 4-μm-thick sections were prepared and stained with the toluidine blue staining. Ultimately, all the samples were air-dried overnight and viewed with a Hitachi Model H-7500 TEM (Hitachi, Japan).
Statistical analysis
Data were shown as mean ± SD, and statistical analysis was performed using SPSS 17.0 software (IBM SPSS, Armonk, NY, USA). Group comparisons were performed using one-way ANOVA followed by a least significant difference (LSD) post-hoc comparisons. P<0.05 was considered to indicate a statistically significant difference.
RESULTS
Identification of human dental pulp cells (hDPCs)
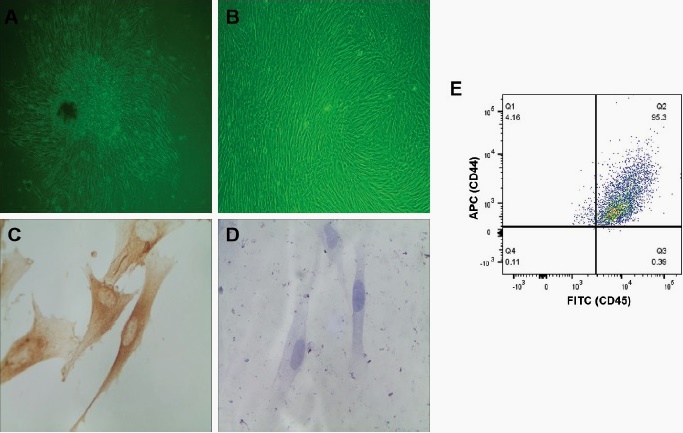
The inverted microscopy showed that hDPCs displayed clustered growth after seven days of adherent culture, primarily with a spindle-shaped morphology, while a smaller fraction of polygonal shapes (Fig. 1A). After passaging, the hDPCs adopted a vortex-like growth pattern, characterized by a more uniform, short spindle-shaped morphology with extending cytoplasmic processes (Fig. 1B). Immunocytochemical analysis was performed using passage 4 hDPCs, and showed positive staining for vimentin (Fig. 1C) and negative staining for cytokeratin (Fig. 1D), indicating their mesenchymal origin. Additionally, the flow cytometry analysis showed that the purity of hDPCs reached more than 95% (Fig. 1E). These morphological features were aligned with the typical biological characteristics of hDPCs.
IGF-1 promoted proliferation of hDPCs in vitro
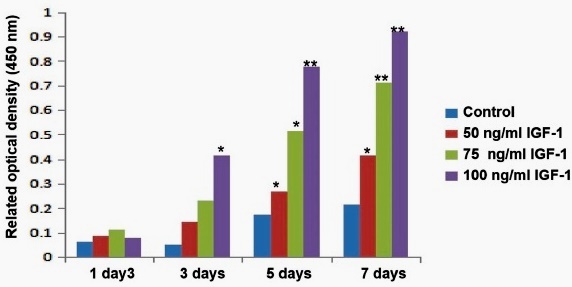
MMT assay was performed to assess the effect of different IGF-1 concentrations (50, 75, and 100ng/mL) on the proliferation ability of hDPCs. As shown in Fig. 2, there was no significant difference among groups (p>0.05), and 100ng/mL IGF-1 significantly increased the proliferation of hDPCs three days after treatment compared to the control group (p<0.01). At day five and day seven, treatment with IGF-1 significantly increased the proliferation of hDPCs compared with the control group (p<0.05 and p<0.01), 100 ng/mL IGF-1 displayed a more substantial enhancement compared to 50 ng/mL and 75 ng/mL (p<0.05 and p <0.01). These results indicated that IGF-1 promoted the proliferation of hDPCs in a concentration-dependent manner.
IGF-1 promoted odontogenic differentiation of hDPCs in vivo
In order to investigate the effect of IGF-1 on osteogenic differentiation in vivo, hDPCs were seeded into an ADM scaffold for the induction of odontogenic differentiation, and the hDPCs-scaffold complexes were transplanted subcutaneously into immunodeficient mice for two or four weeks. HE staining conducted at 2 and 4 weeks post-transplantation revealed more cells and extracellular matrix in implants from IGF-1 treatment group compared to the control group (Fig. 3). Alizarin red staining demonstrated that mineralized nodules in the IGF-1 group exhibited time-dependent enhancement, including the differentiation of odontoblast-like cells (Fig. 4). TEM analysis of hDPCs at 2 and 4 weeks post-transplantation showed an abundance of mitochondria, rough endoplasmic reticulum and Golgi complexes (Fig. 5). These findings indicated that IGF-1 progressively promoted the odontogenic differentiation of hDPCs in vivo.
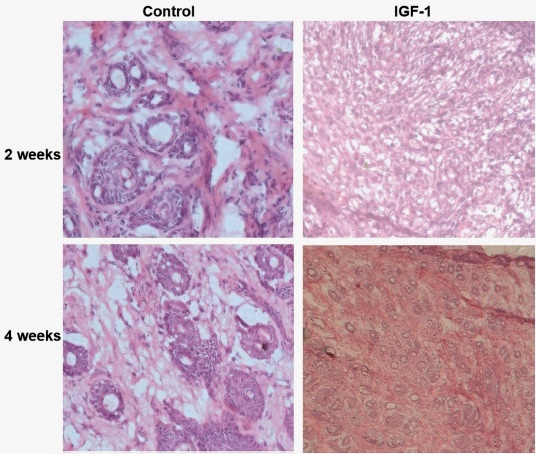
Fig. 3 HE staining revealed more cells and extracellular matrix in implants from the IGF-1 group compared to the control group (without IGF-1) at 2 and 4 weeks post-transplantation.
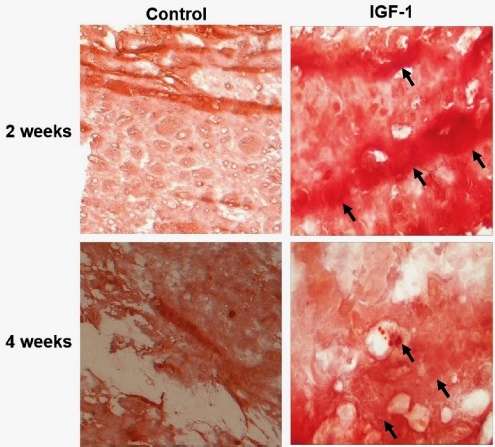
Fig. 4 IGF-1 promoted odontogenic differentiation of hDPCs in vivo. Alizarin red staining was performed to identify calcium-containing odontoblast, showing more mineralized tissues (arrows) in IGF-1 group compared to the control group (without IGF-1) at 2 and 4 weeks post-transplantation.
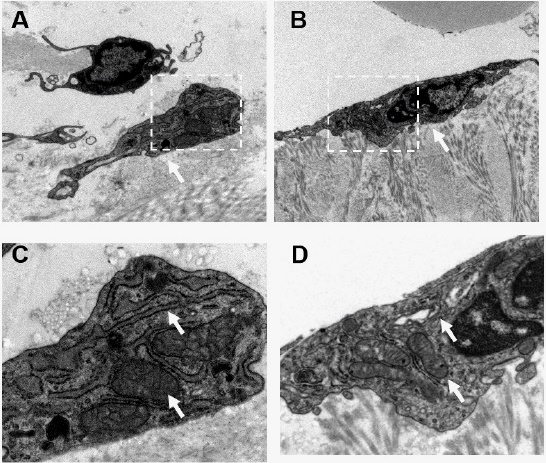
Fig 5 TEM analysis. A-B: The subcellular structure of hDPCs in scaffold 2 weeks (A) and 4 weeks (B) post-transplantation in IGF-1 group (magnification × 6K). C-D: The subcellular structure of hDPCs in scaffold 2 weeks (C) and 4 weeks (D) post-transplantation in IGF-1 group (magnification × 15K). Mitochondria and rough endoplasmic reticulum were highlighted using arrows.
DISCUSSION
Cell-based tissue engineering has become an irreplaceable method for regenerating dental tissues 9, and the potential value of hDPCs has been widely accepted in bone tissue engineering due to their high self-renewal capacity and stemness 22,23. The process of dentinal regeneration involves the proliferation and differentiation of hDPCs into odontoblasts, dental pulp healing, and reparative dentin formation 24. Increasing studies have implicated IGF-1 in the maintenance of proliferation and differentiation of various stem cells, such as embryonic stem cells, bone marrow mesenchymal stem cells (BMSC), periodontal ligament stem cells, and hDPSCs 25. However, the effect of IGF-1 on the proliferation and differentiation of hDPCs in vitro and in vivo warrants further investigation. In this study, we found that exogenous IGF-1 promoted the proliferation of hDPCs in a concentration-dependent manner.
Additionally, IGF-1 progressively promoted odontogenic differentiation of hDPCs in vivo by subcutaneous transplantation into nude mice.
As a unique type of dental stem cell, hDPCs are derived from the pulp in deep carious teeth and display stronger proliferation and osteogenic differentiation capability due to distinctive environmental stimulus 26,27. This distinctive trait provides valuable insights into the cellular mechanisms underlying tooth development and regenerative repair, offering the potential for dental pulp regeneration. The proliferation ability of colony-forming cultures of hDPCs and BMSCs was assessed by bromodeoxyuridine, showing that the number of proliferating cells in hDPC cultures was significantly higher than that in BMSC cultures. This result proves the more substantial proliferation capability of hDPCs compared to BMSCs 28. Moreover, under specific induction conditions, hDPCs can be induced to differentiate into various cell types, including odontoblast-like and osteoblast-like cells. Almushayt et al. 29 experimentally verified the capacity of hDPCs to differentiate into odontoblast-like cells in vitro. This study also found the odontoblast differentiation potential of hDPCs in vivo by subcutaneous transplantation into nude mice. Zhang et al.30 isolated dental pulp cells from healthy third molars through enzymatic digestion, confirming their identity as hDPSCs. Subsequent induction assays revealed their multilineage differentiation potential, including differentiation potential toward adipogenic, osteogenic, fibrogenic, chondrogenic, and neurogenic lineages. Yang et al. 31 demonstrated the capacity of dental pulp stem cells to differentiate into chondrocytes and myocytes under specific induction conditions.
Growth factors are critical in tooth tissue regeneration, as they have essential roles in regulating cell functions, such as platelet- derived growth factor, fibroblast growth factor, and epidermal growth factor 32,33. IGF-1, one of the ubiquitous peptide hormones, has been identified to facilitate various cellular processes, including cell proliferation, differentiation, migration, apoptosis, and survival 25. Previous studies have reported that IGF-1 can affect hDPCs proliferation, promote osteogenesis, and help reconstruct tooth- supporting tissues 34-36. A report by Onishi et al. 37 reveals multiple biological effects of IGF-1 on hDPCs. IGF-1 can stimulate hDPCs proliferation, increase mucin and extracellular matrix protein synthesis, enhance DNA synthesis, and increase alkaline phosphatase (ALPase) activity when hDPCs were cultured in a serum-free medium supplemented with IGF-1. Increasing investigations have illustrated the capacity of IGF-1 to induce the differentiation of ameloblasts and odontoblasts, facilitating dentin regeneration and the development of structures resembling enamel-dentin complexes 34,35. IGF-1 also influences the proliferation and differentiation of other odontogenic cell types, specifically periodontal ligament stem cells and stem cells from apical papilla 19,38. In this study, we also found that exogenous IGF-1 promoted the proliferation of hDPCs in vivo in a concentration-dependent manner and progressively promoted odontogenic differentiation of hDPCs in vivo.
IGF-1 receptor (IGF-1R) is a cell-surface receptor tyrosine kinase with its specific ligands IGF-1 39. It activates downstream signaling pathways, namely the phosphoinositide 3-kinase (PI3K)/AKT and the RAS/ mitogen-activated protein kinase (MAPK) pathways. These pathways are widely involved in stem cell growth, proliferation, and differentiation 40,41. Previous studies have reported that the activation of IGF-1R signaling contributes to maintaining the self-renewal and differentiation of hDPCs 25. It has been demonstrated that treatment with IGF-1 can up-regulate the expression of phospho-ERK and phospho-p38 of hDPCs, suggesting MAPK signaling pathway during differentiation of hDPSCs and human periodontal ligament stem cells 19. The interactions between IGF-1R and the p38 MAPK signaling pathway control the quiescence and activation of hDPSCs. In addition, the downstream effectors between the MAPK signaling pathway and the PI3K/Akt signaling pathway partly overlap, resulting in the interaction effect of two signaling pathways. Some studies show that IGF-1 can promote the differentiation of adipose-derived stem cells and endothelial cells by the PI3K/AKT signaling pathway 42) Meanwhile, the IGF-1-induced activation of the AKT signaling pathway also involved the proliferation, migration, osteogenesis, and angiogenesis of hDPSCs 21. However, the underlying molecular mechanism of proliferation and odontogenic differentiation for hDPCs needs further research.
In conclusion, we found that exogenous IGF-1 promoted the proliferation of hDPCs in a concentration-dependent manner in vitro and progressively promoted odontogenic differentiation of hDPCs in vivo. This suggests that modifying IGF-1 signaling may offer potential strategies for hDPCs-based tissue engineering to regenerate dental tissues.