INTRODUCTION
Arsenic is a metalloid commonly found in the environment, known for its toxicity and important public health effects 1,2. It mainly impacts populations through contaminated drinking water, often from natural sources and human activities such as industrial discharge and the use of arsenic-based pesticides 3,4. According to the World Health Organization (WHO), more than 140 million people worldwide are exposed to arsenic- contaminated water exceeding the safe limit of 10 µg/L 5. Furthermore, long-term exposure to arsenic has been associated with numerous health problems, including cancer and various non-cancerous conditions like cardiovascular diseases, diabetes, and reproductive issues 6,7. Sodium arsenite is recognized for causing testicular toxicity through mechanisms such as oxidative stress, apoptosis, and inflammation. Researchers believe that inflammation, oxidative/nitrosative stress, and apoptosis are key factors in arsenic-induced testicular damage 6,8.
The impact of arsenic toxicity on reproductive health has become an increasing concern, particularly regarding the male reproductive system 9. Arsenic exposure is linked to various reproductive problems, including male infertility, decreased sperm quality, and disruptions in testosterone production. This results in reduced weights of reproductive organs, more sperm abnormalities, and apoptosis in testicular cells 10. The harmful effects of arsenic on male fertility involve several mechanisms, such as interference with steroidogenesis and mitochondrial dysfunction in reproductive tissues 9. These disruptions lead to lower testosterone levels, decreased sperm count, and abnormal sperm morphology 11. Additionally, changes in spermatogenesis and reduced gonadotropin levels have been observed, further emphasizing the reproductive risks of arsenic 3. Moreover, arsenic exposure causes increased production of reactive oxygen species (ROS), indicating the role of oxidative stress in its genotoxic effects 12. Researchers have documented that testicular toxicity caused by sodium arsenite is partly due to its ability to induce apoptosis through the p53 pathway, as shown in zebrafish studies where sodium arsenite exposure elevated apoptosis markers and decreased global DNA methylation 13. Furthermore, experiments with Caenorhabditis elegans demonstrated that sodium arsenite can cause cell cycle arrest and germline apoptosis, both of which depend on concentration and exposure time 12.
Several protective agents have been studied to reduce testicular toxicity caused by sodium arsenite. Melatonin has been shown to decrease arsenic-induced testicular cell death, oxidative stress, and tissue damage by boosting antioxidant enzyme activity and lowering lipid peroxidation 14. Similarly, Salvia hispanica (chia seeds) proved effective in decreasing testicular toxicity by improving sperm quality, serum sex hormone levels, and antioxidant enzyme activity, thanks to its flavonoid content and antioxidant properties 15. Hesperidin and lipoic acid were also tested for their protective effects against liver, kidney, and testicular toxicity when administered with sodium arsenite16. Rutin is a natural flavonoid with a wide range of pharmacological benefits, including antioxidant, antimicrobial, antifungal, anti-allergic, anti-cancer, anti-inflammatory, and antiapoptotic effects 4,17-22. Additionally, rutin has been shown to help manage neurodegenerative diseases and metabolic conditions like diabetes due to its cell-protective effects 19. Its protective properties have been documented in the liver, kidneys, and heart due to its anti- inflammatory, antioxidant, and antiapoptotic actions, making it a candidate for protecting these organs from toxic agents 4. However, the impact of rutin on testicular toxicity caused by sodium arsenite has not yet been studied. Given its protective effects on other organs, rutin might provide similar benefits in reducing testicular damage from sodium arsenite. This study examined the effectiveness of rutin in alleviating testicular toxicity caused by sodium arsenite in male rats.
MATERIALS AND METHODS
Experimental design
Rats (male, Sprague-Dawley, weighing 200-220 g, sourced from the animal house of Shaanxi Kangfu Hospital, China) were randomly divided into the following groups (n=6 per group): normal, arsenic control (As), rutin (Sigma Chemical Co., St Louis, Missouri, United States; 25, 50, and 100 mg/kg), and Co-enzyme Q10 (Medicines Pvt. Ltd. Mumbai, India; 10 mg/kg). Testicular damage was induced in rats (except the normal group) by oral administration of sodium arsenite (Otto Chemicals, India; 10 mg/kg, for two days) 23,24. Rats in the arsenic control and normal groups were treated with doubledistilled water (10 mL/kg). Three different dosages of rutin (25, 50, and 100 mg/kg) were selected based on previous studies 21,22) and were administered orally for 15 days. Coenzyme Q10 was administered for seven consecutive days before arsenite administration and continued for up to 15 days. On the 16th day, the rats were put under anesthesia using ether, and blood was drawn through retro-orbital puncture. Each blood sample was placed in a separate vial for analysis of serum parameters. Serum follicle-stimulating hormone, luteinizing hormone, and testosterone levels were quantified according to the manufacturer’s instructions for the rat-specific ELISA kit (Thermo Scientific, Rockford, IL, USA). The rats were euthanized by carbon dioxide asphyxiation, and the testes were rapidly removed and stored at -80°C for biochemical parameters. Other organs, such as the epididymis and prostate, were isolated and weighed. The epididymal sperm count and motility were determined according to a previously reported method 25.
Biochemical estimation of testis homogenate
Supernatant of the tissue homogenate was employed to estimate lipid peroxidation [malondialdehyde (MDA) content], nitric oxide (NO content), reduced glutathione (GSH), and superoxide dismutase (SOD)] as described previously 26-29. Testicular mitochondrial enzyme activities, including Complex I (nicotinamide-adenine dinucleotide (NADH) dehydrogenase activity), Complex II (succinate dehydrogenase (SDH) activity), Complex III (mitochondrial redox activity), and Complex IV (cytochrome oxidase assay) were estimated according to previously reported methods 30,31.
Testicular interleukins (interleukins (ILs); IL -6 (ERA31RB) and IL -1β (BMS630)) and tumor necrosis factor-alpha (TNF-α; ERA56RB) were quantified in the testis homogenate using Enzyme-linked immunosorbent assay (ELISA) kits (Thermo Scientific, Rockford, IL, USA). Briefly, 500 mg of testis tissue samples were homogenized with a mechanical homogenizer in 5 ml of phosphate- buffered saline at 3000 rpm. The homogenate was centrifuged for 30 min at 20,000 rpm (4°C) in a cryo centrifuge (Eppendorf), and the supernatant was used to determine ILs and TNF-α. Briefly, the quantification of ILs and TNF-α was performed using the Thermo Scientific Rat ILs and TNF-α immunoassay kit, following the instructions provided. The Rat ILs and TNF-α immunoassay was a 4.5 h solid phase designed to measure rat ILs and TNF-α levels. The assay employed a sandwich enzyme immunoassay principle. A monoclonal antibody specific for rat ILs and TNF-α was pre-coated on the microplates. Standards, control, and samples were pipetted into the wells, and the immobilized antibody thus bound any rat ILs or TNF-α present in the sample. After washing away the unbound substance, an enzyme-linked polyclonal antibody specific for rat ILs or TNF-α was pipetted into the microtitre wells. Any unbound antibody was washed off, and then a substrate solution was added to the wells. The enzymatic reaction produced a blue product that turned yellow upon addition of the stop solution. The intensity of the generated color was measured and was proportional to the amount of rat ILs or TNF-α bound in the initial steps. A standard curve was run on each assay plate using recombinant ILs or TNF-α in serial dilutions. The sample values were then read and calculations made according to the standard curve. Values were expressed as means ± S.E.M. The levels of ILs or TNF-α were expressed as units per mg of gastric tissue.
The protein expression of caspase-3 and caspase-9 was assessed by western blot in the testis 32-34. Briefly, the testis was sonicated in Tissue Protein Extraction Reagent (Thermo Fisher Scientific, Inc., Mumbai, Maharashtra, India). The lysates were centrifuged at 10,000 × g for 10 min at 4°C. Protein concentration was determined using a Bicinchoninic Acid (BCA) assay kit (Beyotime Shanghai, China) on ice for 30 min. Equal amounts of extracted protein samples (50 µg) were separated by 10% SDS-PAGE (sodium dodecyl sulfate-polyacrylamide gel electrophoresis) and transferred onto polyvinylidene difluoride membranes. The membranes were blocked with 5% non-fat dry milk at 37°C for 1 hr and incubated overnight at 4°C with the primary antibodies recognizing caspase-3 (ab4051; 1/200 dilution; 31 kDa) and caspase-9 (ab202068; 1/2000 dilution; 46 kDa; Abcam, Cambridge, MA, USA). In addition, an anti-rabbit horseradish-linked secondary antibody (goat anti-rabbit IgG H&L; ab97051) was used, which was incubated at 37°C for 2 hr. Protein bands were visualized using the Chemiluminescent kit (Bio-Rad Laboratories, Inc., Mumbai, Maharashtra, India), and glyceraldehyde 3-phosphate dehydrogenase (GAPDH; EPR6256, ab128915; 1/10000 dilution; 36 kDa) served as the loading control.
Histopathological analysis of testis
Other testis samples were preserved in 10% formalin for 24 hours. These samples underwent dehydration and were immersed in xylene for one hour, repeated three times, followed by treatment with ethyl alcohol at concentrations of 70%, 90%, and 100% for two hours. The infiltration and impregnation process involved treating the samples with paraffin wax twice, with each session lasting 1 hour. The tissue samples were then sliced into sections with a thickness of 3-5µm and stained using hematoxylin and eosin (H&E). The sections were mounted on slides with Distrene Pthalate Xylene (DPX) serving as the mounting medium. A light microscope was used to examine the sections for histopathological characteristics and cell infiltration. The observed changes in histological features were categorized into grades ranging from 0 to 4 according to a previously reported method 35.
Statistical analysis
The data are presented as mean ± standard error of the mean (SEM). GraphPad Prism 5.0 software (GraphPad, San Diego, CA, USA) was utilized for data analysis. Biochemical parameter data were examined using one-way analysis of variance, followed by Tukey’s multiple range test for parametric results, while the Kruskal-Wallis test was used for non-parametric outcomes. A p of less than 0.05 was deemed statistically significant. Correlation coefficients were calculated using a two-sided Fisher’s test.
RESULTS
Attenuation of arsenic-induced alteration in body weight and organ weights by rutin
Table 1 presents the descriptive results for various body and organ weight parameters across the different treatment groups. The arsenic (As) control group had a significant reduction (p<0.001) in organ weights (testes, epididymis, and prostate) and body weight compared with the normal group. CoQ treatment resulted in a significant improvement (p<0.001) in body weight and organ weights compared to the arsenic control group. Rutin (50 and 100 mg/kg) administration also effectively (p<0.01 and p<0.001) and dose-dependently increased the body, testes, epididymis, and prostate weights relative to the arsenic-exposed group.
Table 1 Attenuation of arsenic-induced alteration in body weight, organ weights (testes, epididymis, and prostate), and sperm parameters (count, motility, and viability) by rutin.
| Parameters | Normal | As Control | CoQ (10) | R (25) | R (50) | R (100) |
|---|---|---|---|---|---|---|
| Body weight (gm) | 281.8 ± 2.86 | 217.50 ± 3.73### | 275.70 ± 2.69*** | 214.70 ± 2.80 | 247.00 ± 4.02** | 262.20 ± 2.77*** |
| Testes weight (gm) | 1.79 ± 0.04 | 1.03 ± 0.07### | 1.59 ± 0.04*** | 1.07 ± 0.05 | 1.51 ± 0.06** | 1.68 ± 0.02*** |
| Testes weight/Body weight (×10-3) | 6.34 ± 0.15 | 4.75 ± 0.39### | 5.76 ± 0.18*** | 4.99 ± 0.21 | 6.13 ± 0.33** | 6.41 ± 0.12*** |
| Epididymis weight (gm) | 0.51 ± 0.01 | 0.19 ± 0.02### | 0.45 ± 0.02*** | 0.23 ± 0.01 | 0.33 ± 0.01** | 0.45 ± 0.01*** |
| Epididymis weight/Body weight (×10-3) | 1.80 ± 0.03 | 0.87 ± 0.07### | 1.62 ± 0.07*** | 1.09 ± 0.07 | 1.32 ± 0.04** | 1.73 ± 0.04*** |
| Prostate weight (mg) | 612.30 ± 10.94 | 308.30 ± 9.78### | 560.70 ± 9.02*** | 298.70 ± 11.44 | 478.30 ± 11.78** | 541.70 ± 11.45*** |
| Prostate weight/Body weight (×10-3) | 2.18 ± 0.04 | 1.42 ± 0.05### | 2.03 ± 0.04*** | 1.39 ± 0.04 | 1.94 ± 0.07** | 2.07 ± 0.05*** |
| Sperm count (millions/mL) | 60.17 ± 1.25 | 34.00 ± 0.97### | 56.83 ± 1.30*** | 33.83 ± 0.75 | 45.00 ± 1.21** | 51.83 ± 1.25*** |
| Sperm motility (%) | 71.33 ± 1.05 | 40.33 ± 1.02### | 62.00 ± 1.00*** | 44.33 ± 0.95 | 49.17 ± 1.49** | 59.50 ± 0.67*** |
| Dead sperm (%) | 28.67 ± 1.05 | 59.67 ± 1.02### | 38.00 ± 1.00*** | 55.67 ± 0.95 | 50.83 ± 1.49** | 40.50 ± 0.67*** |
| Abnormal sperm (%) | 12.17 ± 0.60 | 36.50 ± 0.43### | 17.17 ± 0.54*** | 34.17 ± 0.48 | 24.67 ± 0.42** | 19.33 ± 0.76*** |
Data analysis: One-way ANOVA (post-hoc test: Tukey’s multiple range test). Data are reported as mean ± SEM (n=6). Statistically significant compared with ###normal rats, ** and ***as control rats. ### p < 0.001, **p < 0.01 and ***p < 0.001. Arsenic (As), Co-enzyme Q10 (CoQ (10)) and Rutin (R).
Attenuation of arsenic-induced alteration in sperm parameters by rutin
The arsenic control group showed a significant reduction (p<0.001) in cell count and motility compared to the control group. However, the percentage of dead and abnormal sperm was markedly higher (p<0.001) in the arsenic control group than that in the normal group. Treatment with CoQ resulted in significant improvements (p<0.001) in sperm count and motility compared with the arsenic control group. Furthermore, CoQ administration resulted in a more effective (p<0.001) recovery of the percentage of dead and abnormal sperm than arsenic control. Rutin (50 and 100 mg/kg) treatment demonstrated a marked (p<0.01 and p<0.001) and dose- dependent improvement in sperm parameters relative to the arsenic-exposed group (Table 1).
Attenuation of arsenic-induced alteration in serum follicle-stimulating hormone, luteinizing hormone, and testosterone levels by rutin
Exposure to sodium arsenite significantly decreased (p<0.001) the levels of serum follicle-stimulating hormone, luteinizing hormone, and testosterone in the arsenic control group compared with the normal group. Treatment with CoQ and rutin (25 mg/kg) had a minimal impact on serum luteinizing hormone, follicle-stimulating hormone, and testosterone hormone levels. However, rutin (50 and 100 mg/kg) treatments demonstrated effective, dose-dependent increases in all three hormones (p<0.01 and p<0.001, respectively) compared with the arsenic control group (Table 2).
Table 2 Attenuation of arsenic-induced alteration in serum luteinizing hormone, follicle stimulating hormone, testosterone levels and testicular mitochondrial complex activities by rutin.
| Parameters | Normal | As Control | CoQ (10) | R (25) | R (50) | R (100) |
|---|---|---|---|---|---|---|
| Serum parameters | ||||||
| Luteinizing hormone (ng/mL) | 1.88 ± 0.04 | 0.26 ± 0.05### | 0.33 ± 0.03 | 0.36 ± 0.04 | 0.71 ± 0.05** | 1.21 ± 0.04*** |
| Follicle stimulating hormone (ng/mL) | 1.37 ± 0.02 | 0.16 ± 0.02### | 0.15 ± 0.02 | 0.26 ± 0.02 | 0.64 ± 0.02** | 0.92 ± 0.02*** |
| Testosterone (ng/mL) | 3.69 ± 0.04 | 1.41 ± 0.05### | 1.32 ± 0.06 | 1.5 ± 0.06 | 2.39 ± 0.04** | 2.86 ± 0.05*** |
| Testicular parameters | ||||||
| Complex I (nmole of NADH oxidized/min/mg protein) | 33.36 ± 1.66 | 7.41 ± 1.80### | 30.21 ± 1.79*** | 10.07 ± 1.35 | 16.91 ± 1.25** | 28.31 ± 2.10*** |
| Complex II (mmole/mg protein) | 14.35 ± 0.82 | 4.38 ± 0.78### | 11.86 ± 0.84*** | 4.74 ± 0.62 | 9.42 ± 0.67** | 11.24 ± 0.58*** |
| MTT assay (OD at 540 nm) | 0.45 ± 0.03 | 0.17 ± 0.03### | 0.45 ± 0.03*** | 0.21 ± 0.02 | 0.32 ± 0.01** | 0.39 ± 0.02*** |
| Complex-IV (nmol cyto-C oxidized/min/mg protein) | 6319.00 ± 280.30 | 1008.00 ± 212.70### | 5293.00 ± 254.70*** | 1586.00 ± 191.90 | 3224.00 ± 201.30** | 4515.00 ± 342.00*** |
Data analysis: One-way ANOVA (post-hoc test: Tukey’s multiple range test). Data are reported as mean ± SEM (n=6). Statistically significant compared with ###normal rats, ** and ***as control rats. ### p < 0.001, **p < 0.01 and ***p < 0.001. Arsenic (As), Co-enzyme Q10 (CoQ (10)), 3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide (MTT) and Rutin (R).
Attenuation of arsenic-induced alteration in testicular mitochondrial complex activities by rutin
Arsenic exposure led to a significant reduction (p<0.001) in mitochondrial complex activity compared to the normal group. The results presented in Table 2 demonstrate that both CoQ and rutin (50 and 100 mg/ kg) significantly improved mitochondrial function in arsenic-exposed rats. Specifically, rutin (50 and 100 mg/kg) administration led to notable (p<0.01 and p<0.001) and dose-dependent increases in mitochondrial complex activities compared with the arsenic control group. However, rutin (25 mg/ kg) did not show any significant potential to improve mitochondrial function or mitigate arsenic-induced damage.
Attenuation of arsenic-induced alteration in testicular oxido-nitrosative stress by rutin
The results presented in Table 3 show the effects of rutin treatment on testicular oxido-nitrosative stress. Arsenic exposure caused a substantial (p<0.001) decrease in GSH and SOD levels and a marked (p<0.001) elevation in nitric oxide and MDA levels in the arsenic control group compared to the normal group. CoQ treatment significantly (p<0.001) improved GSH and SOD levels and effectively reduced (p<0.001) nitric oxide and MDA levels compared with the arsenic control group. Rutin (25 mg/kg) treatment showed minimal effects in attenuating arsenic-induced elevated testicular oxido-nitrosative stress compared with the arsenic control group. However, rutin (50 and 100 mg/kg) demonstrated marked (p<0.01 and p<0.001) and dose-dependent protective effects, reflected by restored antioxidant status (GSH and SOD levels) and reduced oxidative stress markers (nitric oxide and MDA) to normal levels (Table 3).
Table 3 Attenuation of arsenic-induced alteration in testicular oxido-nitrosative stress and inflammatory markers by rutin.
| Parameters | Normal | As Control | CoQ (10) | R (25) | R (50) | R (100) |
|---|---|---|---|---|---|---|
| SOD (U/mg of protein) | 13.20 ± 1.10 | 5.19 ± 0.93### | 10.66 ± 0.87*** | 5.45 ± 0.61 | 8.91 ± 0.93** | 10.66 ± 0.96*** |
| GSH (µg/mg of protein) | 13.76 ± 0.64 | 3.90 ± 0.62### | 11.04 ± 0.59*** | 5.36 ± 0.50 | 8.31 ± 0.76** | 10.43 ± 0.56*** |
| MDA (nM/mg of protein) | 0.46 ± 0.10 | 3.14 ± 0.18### | 1.97 ± 0.18*** | 3.00 ± 0.27 | 2.11 ± 0.04** | 1.59 ± 0.19*** |
| NO (µg/mg of protein) | 119.00 ± 9.08 | 284.00 ± 13.96### | 143.90 ± 9.97*** | 261.40 ± 13.64 | 222.10 ± 12.14** | 167.90 ± 9.82*** |
| TNF-α (pg/mL) | 157.00 ± 8.15 | 409.70 ± 12.69### | 222.80 ± 14.07*** | 377.20 ± 14.06 | 330.30 ± 8.02** | 253.70 ± 12.06*** |
| IL-1β (pg/mL) | 17.28 ± 3.86 | 72.12 ± 1.85### | 30.57 ± 2.76*** | 68.46 ± 1.62 | 48.95 ± 2.70** | 42.78 ± 2.41*** |
| IL-6 (pg/mL) | 40.50 ± 5.53 | 152.60 ± 5.18### | 59.41 ± 9.32*** | 155.50 ± 6.21 | 130.40 ± 7.87** | 71.78 ± 7.13*** |
Data analysis: One-way ANOVA (post-hoc test: Tukey’s multiple range test). Data are reported as mean ± SEM (n=6). Statistically significant compared with ###normal rats, ** and ***As control rats. ### p < 0.001, **p < 0.01 and ***p < 0.001. Arsenic (As), Co-enzyme Q10 (CoQ (10)), Glutathione (GSH), Interleukin-1 beta (IL-1β), Interleukin-6 (IL-6), Malondialdehyde (MDA), Nitric Oxide (NO), Rutin (R), Superoxide Dismutase (SOD), and Tumor Necrosis Factor-alpha (TNF-α).
Attenuation of arsenic-induced alteration in testicular inflammatory markers activities by rutin
The results in Table 3 show significant differences in inflammatory cytokine levels between the normal and arsenic control groups. The arsenic control group exhibited markedly elevated (p<0.001) levels of ILs (IL -6 and IL -1β) and TNF-α compared with the normal group. CoQ treatment substantially (p<0.001) lowered ILs (IL -6 and IL - 1β) and TNF-α levels compared to the arsenic control group. Rutin (50 and 100 mg/ kg) administration resulted in effective (p<0.01 and p<0.001) and dose-dependent reductions in cytokine levels compared with the arsenic control group. However, a lower dose of rutin (25 mg/kg) did not reduce cytokine levels to levels comparable to those of the arsenic control group, indicating failure to ameliorate arsenic-induced inflammation (Table 3).
Attenuation of arsenic-induced alteration in testicular caspase-3 and caspase-9 protein expressions by rutin
Fig. 1 illustrates the impact of arsenic exposure on markers of apoptosis in the testes, along with their correlation with sperm count. The caspase-3 and caspase-9 protein expression was significantly increased (p<0.001) in the arsenic control group compared with that in the normal group. The substantial elevation in caspase-3 and caspase-9 protein expression was markedly downregulated (p<0.001) by CoQ treatment compared to the arsenic control group. Treatment with rutin (50 and 100 mg/kg) significantly (p<0.01 and p<0.001) and dose-dependently reduced both caspase-3 and caspase-9 relative densities compared to the arsenic control (Figs. 1A and 1B). Furthermore, Figs. 1C and 1D reveal a strong inverse correlation between sperm count and the relative densities of caspase-3 (R 2 = 0.8168, p<0.001) and Caspase-9 (R2 = 0.7075, p<0.01), respectively, indicating that lower sperm counts are associated with increased apoptotic activity.

Fig. 1 Attenuation of arsenic-induced alterations in testicular caspase-3 (A) and caspase-9 (B) protein expression by rutin. Correlation of sperm count with caspase-3 (C) and caspase-9 (D) protein expression. Data analysis: One-way ANOVA (post-hoc test: Tukey’s multiple range test). Data are reported as mean ± SEM (n=6). Statistically significant compared with ###normal rats, ** and ***As control rats. ### p < 0.001, **p<0.01 and ***p<0.001. Correlation coefficients were determined using a two-sided Fisher’s test. Arsenic (As), Co-enzyme Q10 (CoQ (10)), Glyceraldehyde-3-Phosphate Dehydrogenase (GAPDH), and Rutin (R). Inflammatory infiltration (black arrow), necrosis (red arrow), and Leydig cells damage (blue arrow).
Attenuation of arsenic-induced alteration in testicular histopathology by rutin
Fig. 2 illustrates the histopathological changes in rat testicular tissue after exposure to arsenic and their amelioration across different treatment groups. In the normal group (Fig. 2A), testicular architecture appeared normal, with intact seminiferous tubules containing organized spermatogenic cells and healthy Leydig cells, and no evidence of necrosis. However, it showed mild inflammation (indicated by black arrows). In contrast, the arsenic control group (Fig. 2B) demonstrated histological aberrations characterized by severe inflammatory infiltration (black arrows), necrosis (red arrows), vacuolated spermatogenic cells (blue arrows), and Leydig cell damage, indicating arsenic-induced toxicity. The quantitative histopathological score in Fig. 2G supports these visual observations, as the arsenic control group exhibited significantly (p<0.001) elevated scores for inflammatory infiltration, necrosis, Leydig cell damage, and vacuolated spermatogenic cells than the normal group. Treatment with CoQ significantly reduced (p<0.001) these histopathological aberrations compared to the arsenic control group (Fig. 2C), suggesting a protective effect against arsenic- induced testicular injury. The rutin (25 mg/ kg) group showed a modest reduction in these scores, but this was not significantly different from the higher rutin or CoQ doses (Fig. 2D). Treatment with rutin (50 and 100 mg/kg) showed a significant (p<0.01 and p<0.001) reduction in arsenic-induced histological aberrations in the testes, reflected in a decrease in inflammatory infiltration, necrosis, Leydig cell damage, and vacuolated spermatogenic cells compared to the arsenic control group (Figs 2E, 2F, and 2G).
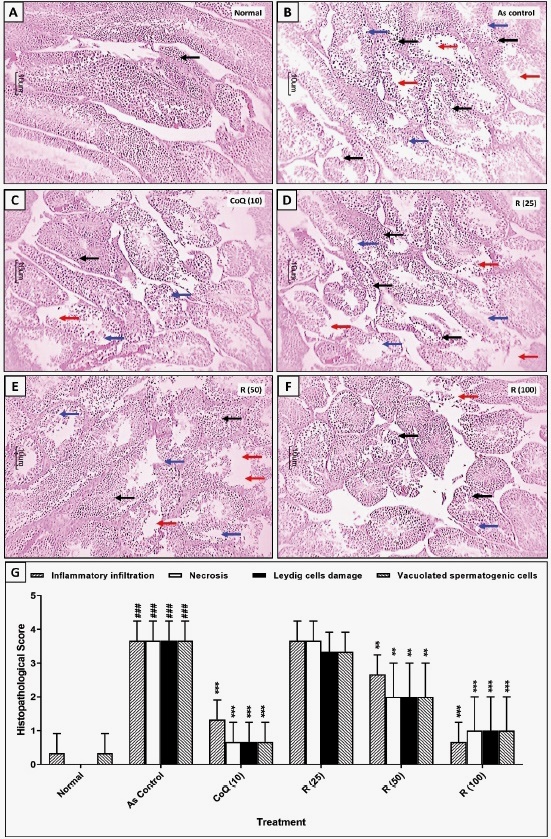
Fig. 2 Attenuation of arsenic-induced alterations in testicular histopathology by rutin treatment. Representative images of testes from each group (A-F). Quantitative analysis of arsenic-induced alterations in testicular histopathology and its attenuation by rutin (G). Data analysis: One-way ANOVA (post-hoc test: Kruskal-Wallis test). Data are reported as mean ± SEM (n=6). Statistically significant compared with ###normal rats, ** and ***As control rats. ### p < 0.001, **p < 0.01 and ***p < 0.001. Arsenic (As), Co-enzyme Q10 (CoQ (10)), Glyceraldehyde-3-Phosphate Dehydrogenase (GAPDH), and Rutin (R).
DISCUSSION
Arsenic exposure has extensive human health effects, with systemic toxicity affecting multiple organs. It is particularly notorious for its carcinogenic properties, causing skin, lung, and bladder cancers, among others 6. However, it is crucial to evaluate its adverse effects on male reproductive health as it directly affects male fertility. Arsenic exposure impairs spermatogenesis and reduces sperm quality through oxidative stress and interference with crucial hormonal signaling pathways 11. Researchers have explored the protective efficacy of phytonutrients, such as lutein and α-lipoic acid, against arsenic-induced oxidative damage in testicular tissues 36. In the present study, rutin, an herbal intervention, was evaluated for its effects on arsenite-induced testicular damage, and the findings suggested that rutin ameliorated testicular toxicity through its anti-apoptotic, anti-inflammatory, and mitochondrial- protective effects in experimental rats.
Integrative proteomic and metabolomic analyses have shown that arsenic exposure significantly alters the proteome and metabolome in rat testes, affecting spermatogenesis and fertilization through disrupted signaling pathways 37. Sodium arsenite causes significant testicular damage primarily through oxidative stress, apoptosis, and inflammatory pathways. Studies have demonstrated that chronic exposure to sodium arsenite can lead to a significant decrease in both absolute and relative testicular weight 38. This reduction in testicular weight is attributed to the toxic effects of arsenic on testicular tissue and its interference with normal spermatogenesis and enzyme activities, including decreased acid phosphatase, sorbitol dehydrogenase, and 17beta-hydroxy-steroid dehydrogenase activities, while increasing lactate dehydrogenase and gamma-glutamyl transpeptidase activities. Additionally, arsenite exposure leads to a notable increase in abnormal sperm forms, along with a decrease in sperm count and motility 38. Furthermore, arsenic is known to accumulate significantly in reproductive tissues, underscoring its potential to cause prolonged toxic effects 38. This accumulation in the testes, epididymis, and prostate is linked to oxidative stress and histopathological alterations, indicating that arsenic’s effects are more profound at the cellular level 8,14. In the present study, administration of rutin significantly attenuated arsenite-induced decreases in testes, epididymis, and prostate weights, suggesting its protective efficacy in reproductive organs.
Sodium arsenite led to decreased levels of serum testosterone, luteinizing hormone, and follicle-stimulating hormone, and to significant changes in sperm parameters, including reduced sperm count and motility, and an increase in abnormal sperm15. Serum hormone levels, such as testosterone, FSH, and LH, play crucial roles in regulating male reproductive function 39. Testosterone, which is primarily produced in the testes, is vital for normal male reproductive functions and secondary sexual characteristics. FSH and LH, secreted by the pituitary gland, regulate testicular function, including steroidogenesis and spermatogenesis 40. Chronic exposure to arsenite is often associated with reduced testosterone levels owing to impaired testicular function. This can be attributed to the direct cytotoxic effects of arsenite on Leydig cells, which are responsible for testosterone production. Additionally, alterations in hormone levels can disrupt the hypothalamic-pituitary-gonadal axis, leading to compensatory changes in LH and FSH levels. In this study, arsenite-induced testicular damage was reflected in changes in serum testosterone, FSH, and LH levels. However, administration of rutin restored the diminished levels of these hormones in the serum.
Oxidative stress plays a crucial role in arsenic-induced toxicity, impacting various biological systems and leading to significant health issues. Arsenic, a toxic metalloid, can cross cellular barriers and accumulate in tissues, contributing to oxidative stress by generating ROS 41,42. ROS generation is central to the pathogenesis of arsenic toxicity, as it leads to mitochondrial dysfunction, a critical factor in neurotoxicity, and other health issues 43,44. Arsenic-induced oxidative stress leads to mitochondrial damage by disrupting the normal balance between antioxidants and pro-oxidants within cells. This disruption impairs mitochondrial membrane potential, promotes cytochrome c release, and triggers apoptosis through caspase activation42. On a systemic level, oxidative stress also causes chromosomal instability and DNA damage, leading to further complications, such as carcinogenesis and neurological disorders44-47. Moreover, arsenic exposure has been shown to deplete antioxidant capacities and elevate oxidative damage biomarkers, such as malondialdehyde (MDA) and nitric oxide, owing to the heightened oxidative environment48,49. These changes underline the oxidative stress mechanism as a cornerstone of arsenic toxicity, which affects various systems, including the reproductive system 50. Therapeutic strategies to combat arsenic-induced oxidative stress focus on enhancing mitochondrial function and increasing antioxidant defense. Acetyl-L -carnitine (ALC), for instance, has been shown to counteract arsenic-induced oxidative stress by improving antioxidant mechanisms and mitochondrial function, thus offering a potential therapeutic pathway 42. Other antioxidants, such as curcumin and apigenin, also exhibit protective effects by reducing ROS and supporting cellular anti-oxidant systems, highlighting their possible roles in mitigating arsenic toxicity51,52. In the present investigation, arsenic-induced toxicity, associated with elevated oxidative stress, contributed to various cellular and systemic damage, as reflected by diminished testicular mitochondrial enzyme activity. However, rutin treatment effectively increased testicular glutathione and superoxide levels and reduced malondialdehyde and nitric oxide levels, suggesting its protective effects against arsenic-induced mitochondrial dysfunction, potentially by mitigating oxidative stress and enhancing mitochondrial energy production.
Inflammation is a significant contributor to arsenic-induced testicular toxicity. Pro-inflammatory cytokines, such as TNF-α and interleukins, mediate this process. TNF-α induces apoptosis and promotes inflammatory responses 53. Inflammation induced by TNF-α can activate various signaling pathways, culminating in oxidative stress and tissue damage 54-56. It has been observed that arsenic exposure results in the upregulation of inflammatory mediators, such as TNF-α and interleukins, contributing to testicular damage 57. Furthermore, Caspases 3 and 9 play crucial roles in the apoptotic pathways underlying arsenic-induced testicular toxicity. Caspase-9 is part of the intrinsic or mitochondrial apoptosis pathway, which is often activated by cellular stressors, including toxins such as arsenic. It is responsible for activating downstream effector caspases, including caspase-3 58,59. Caspase-3 then executes apoptosis by cleaving cellular substrates, leading to systematic breakdown of cellular components and cell death 60-62. In arsenic-induced testicular toxicity, activation of these caspases is indicative of enhanced apoptotic activity, contributing to the observed tissue damage and dysfunction 57. In the current study, arsenic-induced elevated inflammation and apoptosis, mediated by cytokines such as TNF-α and enzymes such as caspases 3 and 9, highlighted the development of arsenic- induced testicular toxicity. However, rutin treatment mitigated the toxic effects induced by chronic exposure to arsenic via the amelioration of these pathways, suggesting its therapeutic potential against arsenic-induced testicular toxicity.
This study has some limitations. First, the study focused on acute arsenic exposure (two consecutive days), which may not reflect the effects of chronic, long-term exposure often observed in human populations. Second, the study only used sodium arsenite, whereas environmental arsenic exposure often involves multiple arsenic species. Third, this study did not address the bioavailability and metabolism of rutin in rats, which could affect its efficacy in humans. Lastly, the study focused on rutin alone and did not explore its potential synergistic effects with other protective agents.
As a conclusion, rutin demonstrated significant protective effects against sodium arsenite-induced testicular toxicity in rats. Rutin ameliorated arsenic-induced damage by reducing oxidative stress, inflammation, and apoptosis in testicular tissue, while improving sperm parameters and hormone levels. These findings suggest that rutin may have therapeutic potential in mitigating arsenic-related reproductive toxicity, although further research is needed to elucidate its mechanisms of action and clinical applicability fully.












 uBio
uBio 


